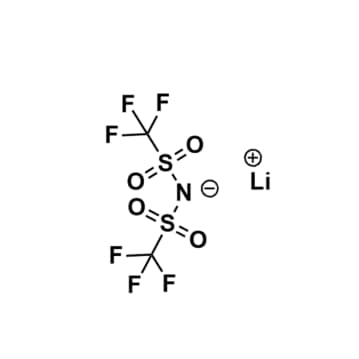
Lithium bis(trifluoromethylsulfonyl)imide, >99%, CAS: 90076-65-6
$117.42 – $2,090.56Price range: $117.42 through $2,090.56
Product Code: KI-0001-HPCAS NO: 90076-65-6
- Chemical Formula: C2F6LiNO4S2
- Synonyms: Li BTA, Li TFSI, Li BTI, Li NTf2
**This product will incur a $97.00 HazMat fee when the order is placed.**
For Battery-grade product, refer to KI-0001-UP
To order 5kg or more, please email us at info@roco.global
Category: Metal Salts
Tags: Battery Materials, CAS: 90076-65-6, lithium salt
SUM Formula: C2F6LiNO4S2
Molecular Weight: 287.19
Melting Point: 236 °C
Purity: >99%
- SUM Formula: C2F6LiNO4S2
- Molecular Weight: 287.19
- Melting Point: 236 °C
Lithium bis(trifluoromethylsulfonyl)imide, CAS: 90076-65-6
Key Applications:
- Lithium‑Ion Batteries – LiTFSI is a key electrolyte salt in liquid and solid electrolytes, offering excellent thermal and electrochemical stability compared to LiPF₆.
- Solid‑State Electrolytes – Used in polymer and ceramic electrolytes, where its large, delocalized anion reduces crystallinity and enhances ionic conductivity.
- Polymer Electrolytes – Incorporated into PEO‑based polymer electrolytes, improving lithium ion transport and mechanical stability for flexible energy devices.
- Supercapacitors – Applied as an electrolyte component in high‑power energy storage systems, leveraging its stability and ionic mobility.
- Electrochemical Research – Studied for ionic conductivity, viscosity, and solvation properties, making it a benchmark salt in thermophysical property investigations.
- Catalysis & Synthesis – Used in anion metathesis reactions to prepare ionic liquids and chiral imidazolium salts.
- Hydrogen Bonding & Solvent Systems – Its weakly coordinating TFSI⁻ anion minimizes ion pairing, enabling stable hydrogen bonding networks in mixed solvents and ionic liquid systems.
- Gas Absorption & Separation – Explored in CO₂ capture studies, where LiTFSI‑based ionic liquids enhance absorption capacity.
Please contact us if you want to learn more or need assistance with your order.
-
Kanchana Jayakody, N., C. Fraenza, C., G. Greenbaum, S., Ashby, D., & S. Dunn, B. (2020). NMR Relaxometry and Diffusometry Analysis of Dynamics in Ionic Liquids and Ionogels for Use in Lithium-Ion Batteries. The Journal of Physical Chemistry B, 124(31), 6843–6856.
-
Widstrom, M. D., Ludwig, K. B., Matthews, J. E., Jarry, A., Erdi, M., Cresce, A. v., Rubloff, G., & Kofinas, P. (2020). Enabling high performance all-solid-state lithium metal batteries using solid polymer electrolytes plasticized with ionic liquid. Electrochimica Acta, 345, 136156.
Related products
-
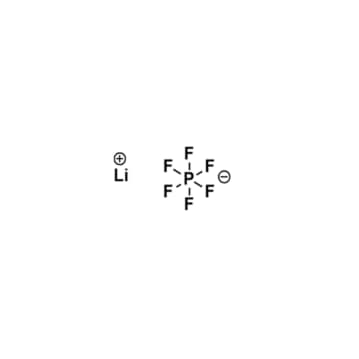
Lithium hexafluorophosphate, battery grade > 99.99%, CAS: 21324-40-3
$401.98 – $2,434.36Price range: $401.98 through $2,434.36Product Code: LBE-0200-HP -
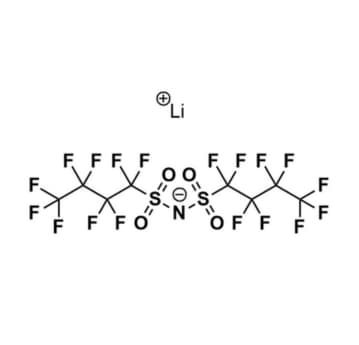
Lithium bis(nonafluorobutanesulfonyl)imide, >98%, CAS: 119229-99-1
$864.77 – $8,850.94Price range: $864.77 through $8,850.94Product Code: KI-0066-HP -
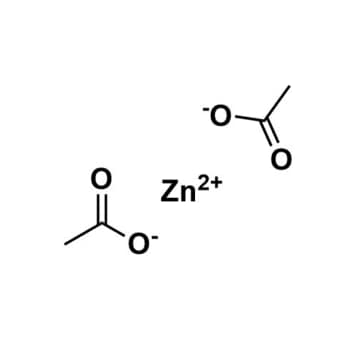
Zinc acetate, >99%, CAS: 557-34-6
$78.50 – $754.60Price range: $78.50 through $754.60Product Code: ROCO-003zn -
Zinc methansulfonate, >98%, CAS: 33684-80-9
Product Code: ZBE-0200-HP