Imidazolium vs. Pyrrolidinium: The Real Trade‑Offs in Ionic Liquid Electrolytes
How molecular structure dictates every trade‑off—and how we can sometimes bend the rules
A technical blog for the curious electrochemist.
Executive Summary:
Imidazolium ionic liquids are fast (low viscosity, high conductivity) but cathodically fragile, while pyrrolidinium ionic liquids are slower but far more resilient at low potential—and formulation choices can shift the balance, but they rarely erase the trade‑off entirely.
Why these two cation families dominate the conversation
At RoCo, we spend a lot of time designing electrolytes—ionic liquids and ionic polymers—for operating conditions where volatility, flammability, and narrow voltage windows are non‑starters. When ionic liquids enter the discussion, two cation families show up again and again: imidazolium and pyrrolidinium. They’re commonly paired with TFSI⁻ (bis(trifluoromethanesulfonyl)imide) because TFSI⁻ tends to deliver high thermal stability and wide anodic stability.
The choice is often presented as a simple pick between “higher conductivity” and “wider electrochemical window.” In practice, that framing is incomplete. The transport penalty (viscosity, conductivity) and the stability penalty (cathodic breakdown pathways) are both direct consequences of molecular structure. Below, we lay out the head‑to‑head numbers, explain the molecular reasons, and then highlight one deeper point: viscosity is not destiny—ionicity can be engineered.
The head‑to‑head: Properties at a glance
Table 1 summarizes practical, experimentally observed ranges for two workhorse ionic liquids: EMIm‑TFSI (1‑ethyl‑3‑methylimidazolium) and BMP‑TFSI (N‑butyl‑N‑methylpyrrolidinium). Values vary with water content, salt loading, temperature, and electrode material, but the ordering is robust.
| Property | EMIm‑TFSI | BMP‑TFSI | Winner |
| Melting point (°C) | −15 to −17 | −18 to −20 | ≈ Tie |
| Viscosity at 25 °C (cP) | ~32–38 | ~75–95 | Imidazolium |
| Ionic conductivity (mS cm⁻¹) | ~8–11 | ~2–4 | Imidazolium |
| Practical electrochemical window (V) | ~4.0–4.5 | ~5.5–6.0 | Pyrrolidinium |
| Cathodic limit vs. Li/Li⁺ | ~1.0–1.2 V (NHC/carbene pathway) | ≈ 0 V (Li plating possible) | Pyrrolidinium |
| Li⁺ transference number* | ~0.1–0.2 | ~0.08–0.15 | ≈ Tie |
| Thermal stability, T₅% (°C) | ~380–420 | ~380–410 | ≈ Tie |
*Strongly dependent on salt concentration, temperature, and measurement method.
The pattern is immediate: imidazolium wins on transport (lower viscosity, higher conductivity), while pyrrolidinium wins on cathodic robustness and therefore usable voltage window for lithium metal. That’s the “no free lunch” principle in electrolyte design.
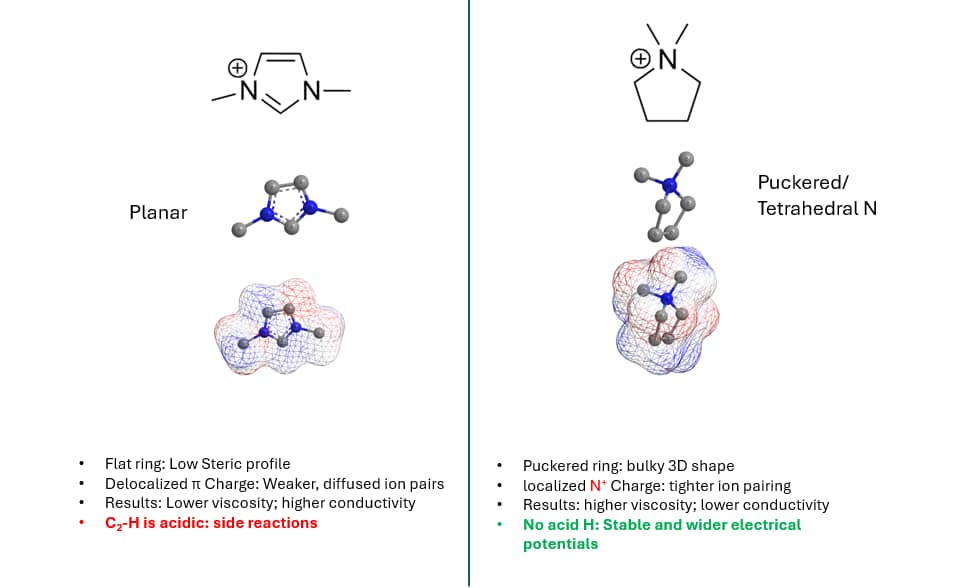
Why imidazolium is faster: aromaticity, planarity, and weaker ion correlation
Imidazolium is a five‑membered aromatic ring with a delocalized positive charge. Two practical consequences follow. First, the cation is relatively planar, which tends to reduce steric resistance to translation and rotation in the liquid. Second, charge delocalization can weaken the persistence of tight contact ion pairs with TFSI⁻, improving mobility. Together, these effects drive lower viscosity and higher conductivity.
Pyrrolidinium cations are saturated rings with a more localized positive charge on nitrogen and a puckered, three‑dimensional geometry. That increases effective hydrodynamic volume and strengthens ion–ion correlations, so viscosity rises and conductivity falls. In batteries, that shows up as higher polarization—unless you compensate with temperature, electrode architecture, or formulation.
The carbene problem: Why many imidazoliums fail before you reach anode potential
The same imidazolium chemistry that improves transport creates a well‑known cathodic failure mode. The C2–H position (between the two nitrogens) is relatively acidic. Under sufficiently reducing conditions, it can participate in reactions that generate N‑heterocyclic carbene (NHC) character, triggering electrolyte decomposition and resistive interphase formation. Practically, that often limits EMIm‑TFSI to ~1.0–1.2 V vs. Li/Li⁺ and makes it a poor fit for lithium metal anodes.
Pyrrolidinium cations have no aromatic C2–H analogue. Their cathodic stability extends close to 0 V vs. Li/Li⁺, so lithium plating and stripping are electrochemically accessible. That does not imply chemical inertness: BMP‑TFSI can still be reduced on reactive surfaces, and SEI formation still occurs. The difference is that the electrochemical window is wide enough to operate a lithium metal system with manageable interphase chemistry.
Blocking the C2 position in imidazolium (e.g., C2‑methylation) suppresses the carbene pathway, but introduces new trade‑offs. Viscosity typically increases and ionicity can decrease as local structure and ion association change. You gain chemical stability but often surrender part of the transport advantage that drew you to imidazolium in the first place.
Beyond viscosity: Why the Walden rule is incomplete
A standard heuristic in ionic liquids is the Walden rule: conductivity scales inversely with viscosity. As a first pass, it explains a lot—especially the imidazolium vs. pyrrolidinium contrast. But it also hides a second control knob: ionicity.
Viscosity tells you how fast species can move. Ionicity tells you what fraction of that motion actually carries charge (as opposed to moving as neutral or weakly charged pairs/clusters). From an engineering perspective, this distinction matters because it determines whether conductivity losses are fundamentally structural or merely kinetic—and only the latter can be recovered through temperature or formulation changes.
Lessons from triazoliums: Tuning ionicity without “just lowering viscosity”
In our published work on 1,2,3‑triazolium ionic liquids, we found clear examples where small structural changes shift ionicity enough to move conductivity beyond what viscosity alone would predict. In one study, two regioisomers with identical formulas (same mass, same anion) showed markedly different Walden behavior. The difference was not “how long the chain was,” but where that chain sat relative to the charged center—changing steric accessibility and the persistence of ion pairing.
The takeaway is not that triazolium is a magic cation. It’s that structure–property relationships in ionic electrolytes are constraints, not commandments. Regioisomerism, branching, and local sterics can tune ion association and effective charge transport—sometimes letting you bend the usual viscosity–conductivity trade‑off.
What this means for electrolyte design (ionic liquids and ionic polymers)
- If your anode never approaches lithium metal potentials (e.g., many intercalation systems), imidazolium‑TFSI can offer useful conductivity and anodic stability—so long as you stay away from the cathodic failure mode.
- If you need operation near 0 V vs. Li/Li⁺ (lithium metal, aggressive anode chemistries), pyrrolidinium‑TFSI is the more robust starting point, and you manage viscosity via temperature, salt selection, or architecture.
- If you’re building ionic polymers or gelled ionic systems, the same logic applies: segmental motion sets the kinetic ceiling, but ionicity (how “free” the charge carriers are) often determines real conductivity.
How we think about this at RoCo
The imidazolium–pyrrolidinium comparison is a canonical example of an engineering trade‑off that starts at the molecular scale. Imidazolium buys transport through aromaticity and charge delocalization, but pays for it with cathodic fragility. Pyrrolidinium removes that failure mode, but accepts higher viscosity and lower conductivity. Neither is “better” in the abstract; the right choice depends on voltage window, electrode chemistry, and operating conditions.
Where the field still has room to grow—and where we focus—is in deliberately breaking assumed couplings between viscosity, ionicity, and stability. If you know where they break, you can design electrolytes that operate intentionally at (or just beyond) those boundaries.
Electrolytes (Ionic Liquids and Ionic Polymers). Further Reading and Primary Sources
(1) Watkins, T.; Roth, E. A.; Lartey, M.; Albenze, E.; Zhong, C.; Luebke, D. R.; Nulwala, H. B. Ionic Liquid Regioisomers: Structure Effect on the Thermal and Physical Properties. New J. Chem. 2015, 39, 1563–1566. https://doi.org/10.1039/C4NJ02044G.
(2) Lartey, M.; Meyer-Ilse, J.; Watkins, T.; Roth, E. A.; Bowser, B.; Kusuma, V.; Damodaran, K.; Zhou, J.; Haranczyk, M.; Albenze, E.; Luebke, D. R.; Hopkinson, D.; Kortright, J.; Nulwala, H. B. Branched Isomeric 1,2,3-Triazolium-Based Ionic Liquids: New Insight into Structure–Property Relationships. Phys. Chem. Chem. Phys. 2015, 17, 29834–29843. https://doi.org/10.1039/C5CP04630A.
(3) Nulwala, H. B.; Tang, S.; Kail, B. W.; Damodaran, K.; Kaur, P.; Wickramanayake, S.; Shi, F.; Luebke, D. R. Probing the Structure–Property Relationship of Regioisomeric Ionic Liquids with Click Chemistry. Green Chem. 2011, 13, 3345–3349. https://doi.org/10.1039/C1GC15721F.
