Phosphonium-based ionic liquids (ILs) are a high-performance class of molten salts valued for their exceptional stability and tunability. By replacing the typical nitrogen-based cation (as in imidazolium or ammonium ILs) with a quaternary phosphonium cation, these ILs tends to have greater thermal and chemical robustness, lower volatility, and flexible polarity – a combination that lets them outperform more common ILs when it comes to thermal stability.
Below, we concisely explore what makes phosphonium ILs stand out, how they compare to other IL families, their key application areas, and a quick look at RoCo®’s top five phosphonium IL products with their properties and uses:
Why Phosphonium ILs Are Considered “High-Performance”
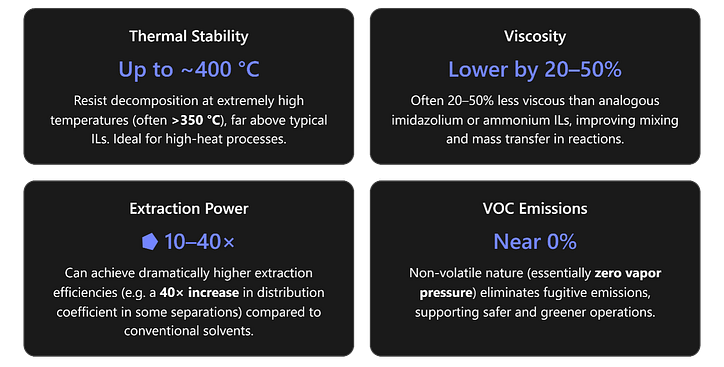
- HigherStability: Phosphonium ILs are thermally and chemically sturdier than their imidazolium or ammonium counterparts. They lack acidic protons, so they withstand strongly basic conditions that would degrade imidazolium ILs (which have an acid-sensitive C2 hydrogen). In thermal terms, many phosphonium ILs remain intact well beyond 300 °C (with decomposition often around 350–400 °C in TGA tests), whereas comparable ammonium ILs break down at temperatures roughly 100 °C lower. This superior stability means phosphonium ILs can handle high-temperature reactions and prolonged operation without breaking apart or losing effectiveness.
- Tunable Polarity & Hydrophobicity: By choosing different alkyl chains on the phosphonium cation and pairing with various anions, we have fine-tuned phosphonium IL properties from polar (even hydrogen-bond donating) to highly hydrophobic. Notably, many common phosphonium ILs are more hydrophobic than imidazolium ILs. For example, phosphonium ILs with bulky organic anions (like phosphinates or fluorinated anions) are essentially immiscible with water, forming dedicated nonaqueous phases – perfect for biphasic systems such as liquid–liquid extractions or two-phase catalysis. On the other hand, phosphonium ILs with polar anions (e.g., methylsulfate) can dissolve polar substrates and even facilitate hydrogen-bonding interactions, acting as powerful solvents for biomass and polymers processing. This solvation is a key advantage: phosphonium ILs can be tailored to “like dissolves like” for a given task better than most solvent systems.
- Lower Viscosity, Better Fluidity: A practical edge of phosphonium ILs is their tendency toward lower viscosity compared to other ILs of similar molecular weight. The large tetraalkylphosphonium cation has a more diffuse charge and less hydrogen bonding, resulting in weaker intermolecular forces. In practice, a phosphonium IL can be significantly more fluid – often on the order of tens of centipoise less – than an imidazolium analog. This reduced viscosity eases stirring, pumping, and mass transport. In processes like extraction or when used as electrolytes, such improved fluidity can translate to faster diffusion and reaction rates, helping overcome what is sometimes a limitation of ILs (some ILs are so viscous they slow down mixing or separations). It’s worth noting that viscosity differences narrow at elevated temperatures (since all ILs become more fluid when heated), but at room temperature, the phosphonium ILs often hold this advantage.
- Wide Electrochemical Window & Conductivity: Phosphonium ILs also shine in electrochemical stability. They often support a wider electrochemical window (i.e., range of potentials without decomposition) than comparable ammonium ILs. For instance, with non-coordinating anions like PF₆⁻ or NTf₂⁻, a phosphonium IL can typically withstand ~5 V between its oxidation and reduction limits, enabling use in high-voltage electrochemical applications. Meanwhile, they exhibit decent ionic conductivity (often a few mS/cm at room temp. for neat ILs), and even higher when a mobile charge carrier (like Li⁺) is added, making them suitable as electrolyte media. However, it is important that using a phosphonium is not a guarantee that it will result in the ideal electrolyte composition. Importantly, they are non-flammable and remain liquid over a broad temperature range, improving the safety of batteries or supercapacitors that use them.
Comparison: In summary, phosphonium ILs vs. imidazolium/ammonium ILs: More robust (thermally and chemically), often less viscous, tunable from polar to hydrophobic, and wider electrochemical stability, at the slight expense of a larger cation that can sometimes mean lower equivalent conductivity per ion (due to size). They don’t engage in problematic side reactions (no carbene formation as in imidazoliums under strong base, no Hofmann elimination as in quaternary ammoniums). These distinctions make phosphonium ILs a very good candidate for extreme conditions and long-lasting performance where other ILs might falter.
Key Applications and Sustainability Benefits
Leveraging the above traits, phosphonium ILs have proven especially effective in several areas of chemistry and chemical engineering:
- High-Temperature Catalysis: Because of their thermal stability, phosphonium ILs serve as excellent solvents or co-catalysts in high-temperature reactions – e.g. hydrothermal conversions, catalytic pyrolysis, or continuous flow processes that run at 200–250 °C. They won’t evaporate or decompose during the reaction, meaning they can provide a stable liquid medium for reactions that traditional solvents (or less stable ILs) simply couldn’t tolerate. Additionally, in phase-transfer catalysis scenarios, phosphonium ILs (such as tetrabutylphosphonium bromide) can replace classic ammonium salts (like Aliquat® 336), offering the same transfer of reagents between phases but with higher thermal durability. This allows reactions (like substitutions or condensations) to be driven faster and even to completion – studies have reported achieving 100% selectivity in certain O-alkylation reactions using a phosphonium IL as the PTC. Moreover, the catalyst/IL phase is easily recycled after reaction (as the IL remains as a separate phase), aligning with green chemistry practices.
- Separation Processes (Extraction): Phosphonium ILs are widely used in liquid-liquid extractions and separations for both inorganic and organic targets. Their hydrophobic members (like trihexyltetradecylphosphonium with appropriate anions) can efficiently extract metal ions (e.g. Co²⁺, Ni²⁺, rare earths) from aqueous feed into an IL phase, often achieving very high single-stage extraction percentages (near-complete removal of target metals). In one case, a specially formulated phosphonium IL achieved a distribution coefficient >40 for lactic acid extraction, versus ~1 using a conventional amine extractant – illustrating an orders-of-magnitude improvement in extraction power. These ILs enable processes like greener hydrometallurgy, where toxic volatile solvents are replaced by reusable IL phases. They also excel in extracting non-polar organics or pollutants from water, functioning as durable, non-volatile extractants that can be regenerated. Furthermore, because phosphonium ILs form biphasic systems readily (due to their hydrophobicity), they are a cornerstone in biphasic catalysis: a catalyst dissolved in the IL can facilitate a reaction in contact with a second phase (aqueous or organic), and then products can be separated by simple phase decanting. This simplifies purification and allows IL+catalyst reuse across cycles, cutting down waste.
- Electrochemistry & Energy: In advanced batteries and supercapacitors, phosphonium ILs act as safe, high-performance electrolytes. Their wide electrochemical window and non-flammability add safety and stability to devices. For example, phosphonium ILs with anions like NTf₂⁻ are used as electrolytes for lithium-ion batteries capable of operating at elevated temperatures with less risk of thermal runaway. They have been used in metal plating, fuel cells, and electrochemical CO₂ reduction, where their stability allows reactions at extreme potentials or in reactive environments. A notable feature is the ability of some phosphonium ILs to stabilize reactive species: the abstract above notes that even superoxide (O₂⁻) can be generated and stabilized in certain ILs, which is normally very challenging and opens up novel chemistry for metal-air batteries or oxidation reactions. In summary, whenever an electrolyte or electrochemical medium is needed that won’t evaporate, won’t catch fire, and can sustain harsh conditions, phosphonium ILs are strong candidates.
- Materials Processing (Polymers & Biomass): The solvent capability of phosphonium ILs extends to dissolving otherwise stubborn solids. Biomass processing is a prime example: ILs like tributylmethylphosphonium methylsulfate can dissolve significant amounts of cellulose and lignin – up to a substantial weight percentage – thereby enabling the breakdown of plant matter into fine solutions for biofuel or biopolymer production. After dissolution, the cellulose can be precipitated or processed further, and the IL recycled (sometimes with anti-solvents or by phase changes). This has made phosphonium ILs key players in developing recyclable solvent systems for biomass pretreatment, replacing traditional, less efficient methods. In the realm of synthetic polymers, phosphonium ILs can act as plasticizers or processing aids: for instance, adding a small amount of a low-viscosity phosphonium IL can improve the flow of polymer melts or can imbue conductivity to polymer electrolytes (useful for solid-state batteries). They’ve even been formulated into lubricants and tribological fluids, taking advantage of their thermal stability and slipperiness to reduce friction in extreme conditions. Overall, the ability to dissolve, mobilize, or modify materials that are challenging for conventional solvents (like cellulose or highly non-polar polymers) is a standout benefit of phosphonium ILs in materials science.
- Sustainability Perks: Aside from performance, phosphonium ILs contribute to greener chemistry in several ways. Their near-zero vapor pressure means negligible VOC emissions – an IL can often replace a volatile organic solvent and thereby cut out a major source of air pollution and health hazard. This has a concrete impact: for example, an extraction or reaction using an IL emits virtually no fumes, whereas an equivalent process with hexane or dichloromethane would. Moreover, phosphonium ILs are typically recyclable; after use, they can be recovered (by distilling off products or by washing) and reused in subsequent runs. Industrial trials have shown that with proper setup, IL losses can be minimized and the same batch reused many times, dramatically reducing the total waste generation. They also enable energy-efficient process redesign: one can often carry out reactions and separations in ILs at mild conditions (because the IL can hold a catalyst or reagent in solution, or eliminate steps) that would otherwise require more energy or yield more waste. Finally, ongoing research indicates that many phosphonium ILs can be made with lower toxicity and biodegradability in mind (for instance, by selecting appropriate anions). All these factors position phosphonium ILs as tools for not only boosting performance but also for aligning with sustainability and green chemistry goals. [link.springer.com] [cdn.intechopen.com]
RoCo®’s Top 5 Phosphonium IL Products
RoCo® is a leading supplier of high-purity ionic liquids, including a suite of phosphonium ILs. Here are five of our most popular phosphonium IL products, each with a glimpse of its technical profile and use cases:
- Trihexyltetradecylphosphonium Bis(2,4,4trimethylpentyl)phosphinate (Product code: IN-0009-TG) – A hydrophobic, thermally sturdy IL (liquid up to ~350 °C+) often chosen for metal extraction and biphasic catalysis. Its large organic phosphinate anion imparts low water miscibility (great for separating aqueous/organic phases) and can coordinate with metals, enabling the extraction of ions like Co²⁺, Cu²⁺, etc., with very high efficiency. This IL (analogous to the commercial Cyphos® IL 104) has been used in continuous processes and even in the production of critical metals, thanks to its robustness. (Purity ≥90%.)
- Trihexyltetradecylphosphonium Hexafluorophosphate (Product code: IN-0012-TG) – A general-purpose phosphonium IL with the non-coordinating PF₆⁻ anion. It is chemically inert and highly hydrophobic, with very low water uptake (virtually no water dissolves in it). This IL finds use in organic synthesis (as a non-polar solvent) and in electrochemistry – for example, as part of electrolyte formulations where its wide electrochemical window and stability improve performance. It’s also handy for the separation of non-polar compounds or for creating IL biphasic systems combined with water or polar solvents. (Purity ≥95%.)
- Trihexyltetradecylphosphonium Dicyanamide (Product Code: IN-0010-TG) – Notable for its lower viscosity relative to other large phosphonium ILs (the dicyanamide [DCA] anion is small and reduces overall fluid resistance). This IL remains liquid at room temperature with moderate viscosity, improving handling. It’s widely used in polymer processing – for instance, dissolving or blending plastics and cellulose – and in lubricant research (providing an ionic medium with less drag for tribological testing). The DCA anion can also engage in coordination, making this IL useful in certain electrochemical contexts (e.g., as part of ionic conductive gels or in double-layer capacitors). (Purity ≥95%.)
- Tributylmethylphosphonium Methylsulfate (Product Code: IN-0013-TG) – A polar, hydrogen-bond-capable IL that is solid just below room temperature (melting ~34 °C) but used in melt form. It has a relatively higher viscosity (~409 cP at 25 °C) due to strong ion pairing, but this correlates with excellent solvating power for biomass and polar substrates. It can dissolve substantial amounts of cellulose and is thus employed in biomass pretreatment for biofuels or biopolymer production. In catalysis, it serves as a reaction medium for acid-catalyzed and enzymatic reactions that require a polar environment, often boosting yields by improving substrate solubility. (Purity ≥95%.)
- Tetrabutylphosphonium Bromide (Product Code: IN-0014-TG) – A classic quaternary phosphonium salt, typically shipped as a solid (mp ≈100 °C) that forms an IL when melted or dissolved. It’s a versatile phase-transfer catalyst, frequently used to accelerate reactions where anions need to move between aqueous and organic phases (e.g., halogen exchange, Williamson ether synthesis). Compared to analogous ammonium bromides, it offers higher thermal stability and can be recycled more times. Additionally, this salt is a common precursor for other ILs: through metathesis, its bromide can be exchanged with other anions to synthesize custom phosphonium ILs. (Purity ≥98%.)
Summary Table – Key Properties of RoCo®’s Top 5 Phosphonium ILs:
| Phosphonium IL (Code) | Key Properties | Typical Applications |
| Trihexyltetradecylphosphonium bis(2,4,4-trimethylpentyl)phosphinate (IN-0009-TG) |
Hydrophobicity: Extremely high (water immiscible) Thermal stability: ~350 °C+ Coordinating anion: Yes (phosphinate) |
– Metal extraction (e.g. Co, Ni, rare-earth recovery) – Biphasic catalysis (IL phase for catalyst, easy product separation) |
| Trihexyltetradecylphosphonium hexafluorophosphate (IN-0012-TG) |
Hydrophobicity: High (very low water uptake) Electrochemical window: Wide Viscosity: Moderate (less than imidazolium PF₆ analog) |
– Organic solvent replacement (non-volatile medium for organic reactions) – Electrolyte component (stable, non-flammable electrolyte for batteries/supercapacitors) – Hydrophobic separations (extracting non-polar compounds) |
| Trihexyltetradecylphosphonium dicyanamide (IN-0010-TG) |
Viscosity: Lower than other P₄₄₄₁₄ ILs (more fluid) Conductivity: Good ionic mobility Anion functionality: DCA offers mild coordination |
– Polymer & biomass dissolution (e.g. processing cellulose, plastics blends) – Lubricants & heat-transfer fluids (ionic medium for high-temp lubrication studies) – Electrochemical uses (ionic liquid gels, capacitors requiring low-viscosity IL) |
| Tributylmethylphosphonium methylsulfate (IN-0013-TG) |
Polarity: High (protic IL with –SO₄CH₃) Dissolution power: Dissolves polar biopolymers (cellulose, etc.) Viscosity: 409 cP @25 °C (melts at 34 °C) |
– Biomass pretreatment (dissolving cellulose & lignin for biofuel production) – Polar reaction medium (supports acid catalysts, enzymatic reactions in IL) – Ion exchange resin (used in supported IL phases for separations) |
| Tetrabutylphosphonium bromide (IN-0014-TG) |
Phase-transfer catalyst (PTC): Yes (classic PTC bromide) Stability: High (no Hofmann elimination) Melting point: ~100 °C (usable as molten salt) |
– Phase-transfer catalysis (facilitates SN2 and other biphasic reactions, often faster rates than without PTC) – IL synthesis precursor (source for P⁺₄₄₄₄ cation in making other ionic liquids via anion exchange) – Catalyst recycling (immobilizing catalysts in IL phase for reuse) |
Each of these ILs comes with a Certificate of Analysis (CoA), and a Technical datasheet is available.
In conclusion, phosphonium ionic liquids offer a compelling blend of performance (with their stability, versatility, and efficiency gains) and sustainability (through reduced emissions and reusability). They exemplify how innovative chemistry can create solvents that meet the challenges of modern R&D and industrial processes. As you consider solvents and reaction media for your next project, especially one aiming for high impact and green credentials, phosphonium ILs – including the top picks from RoCo® – should be high on your list of options. These liquids are not just alternatives; in many cases, they are enablers of new chemistry, allowing processes to operate in ways not possible before. That is why phosphonium ionic liquids have earned their reputation as high-performance materials in the push toward cleaner, more efficient chemical technologies.
References:
Explore our full range of phosphonium ionic liquids and buy ionic liquids online from a trusted ionic liquid supplier with a proven track record in government, industrial, and institutional deliveries.