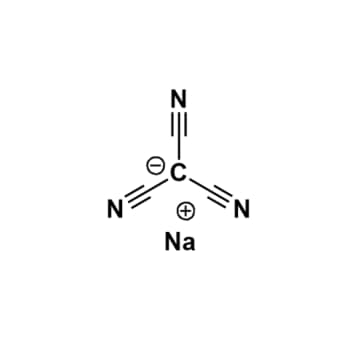
Sodium tricyanomethanide, >98%
$324.10 – $4,553.24Price range: $324.10 through $4,553.24
Product Code: KI-0049-SGCAS NO: 36603-80-2
- Chemical Formula: C4N3Na
- Synonyms: Na TCM
- SMILESC(#N)[C-](C#N)C#N.[Na+]
**This product will incur a $97.00 HazMat fee when order is placed**
Category: Metal Salts
SUM Formula: C4N3Na
Molecular Weight: 113.05
Purity: >98%
- SUM Formula: C4N3Na
- Molecular Weight: 113.05
Sodium tricyanomethanide, CAS: 36603-80-2
Key Applications:
Organic Synthesis & Functional Group Construction
- Serves as a versatile nucleophile for forming C–C and C–N bonds in heterocycle synthesis.
- Enables tricyanomethylation of electrophiles, expanding access to electron‑poor intermediates and push–pull molecular frameworks.
- Used in the preparation of highly conjugated building blocks for dyes, ligands, and donor–acceptor systems.
Coordination Chemistry & Metal Complex Formation
- Acts as a stabilizing anionic ligand for transition‑metal complexes due to its strong electron‑withdrawing character.
- Supports the synthesis of charge‑transfer complexes and metal–organic species with tunable redox properties.
- Useful in studying metal–ligand bonding in systems requiring delocalized, resonance‑stabilized anions.
Materials Science & Electronic Applications
- Incorporated into organic electronic materials, including semiconductors and conductive polymers, to modulate charge density and electron mobility.
- Used in the design of nonlinear optical materials and chromophores due to its strong π‑acceptor behavior.
- Supports development of photoactive and electroactive materials, including radical precursors and redox‑active frameworks.
Mechanistic & Physical Organic Chemistry
- Employed as a model anion for studying resonance stabilization, anion delocalization, and substituent effects.
- Useful in probing reaction kinetics and electron‑transfer mechanisms in systems sensitive to anion structure.
Precursor for Advanced Cyanocarbon Derivatives
- Functions as a starting material for synthesizing tetracyanoethene (TCNE) analogs, cyanocarbon clusters, and other electron‑poor scaffolds.
- Enables access to high‑energy density molecules and strong acceptor units used in molecular electronics.
Click‑Reaction Catalysis & Reactive Intermediate Generation
- Supports anion‑mediated activation pathways relevant to click‑type transformations, particularly in systems requiring strong π‑acceptor anions.
- Facilitates nucleophile‑driven cycloaddition chemistry, enabling formation of electron‑poor heterocycles and modular building blocks.
- Useful in developing metal‑free or low‑metal‑loading click strategies, where the tricyanomethanide anion enhances reactivity through charge delocalization and electrophile polarization.
-
Królikowski, M., Więckowski, M., Ebrahiminejadhasanabadi, M., Nelson, W. M., Naidoo, P., Ramjugernath, D., & Domańska, U. (2022). Carbon dioxide solubility in ionic liquids: [Guad-(6,6),(1,1),(1,1)][DCA] and [Guad-(6,6),(1,1),(1,1)][TCM] at high pressure. Fluid Phase Equilibria, 563, 113572.
-
Łukomska, A., Wiśniewska, A., Dąbrowski, Z., Kolasa, D., Luchcińska, S., & Domańska, U. (2021). Separation of cobalt, lithium and nickel from the “black mass” of waste Li-ion batteries by ionic liquids, DESs and organophosphorous-based acids extraction. Journal of Molecular Liquids, 343, 117694.
-
Królikowski, M. (2019). Liquid–liquid extraction of sulfur compounds from heptane with tricyanomethanide based ionic liquids. The Journal of Chemical Thermodynamics, 131, 460–470.
You may also like…
-
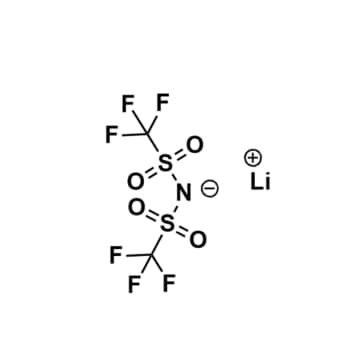
Lithium bis(trifluoromethylsulfonyl)imide, Battery Grade
$154.26 – $1,222.85Price range: $154.26 through $1,222.85Product Code: KI-0001-UP -
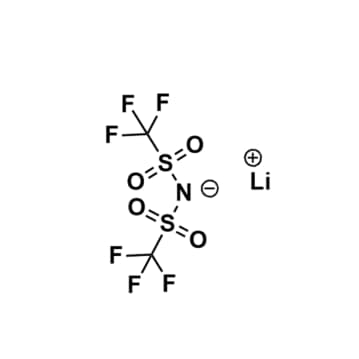
Lithium bis(trifluoromethylsulfonyl)imide, >99%
$117.42 – $2,090.56Price range: $117.42 through $2,090.56Product Code: KI-0001-HP -
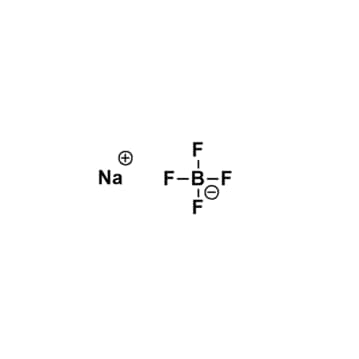
Sodium tetrafluoroborate, >98%
$56.53 – $215.72Price range: $56.53 through $215.72Product Code: KI-0025-HP -
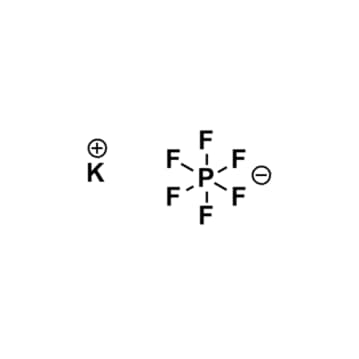
Potassium hexafluorophosphate, >99%
$120.27 – $1,058.58Price range: $120.27 through $1,058.58Product Code: KI-0031-HP
Related products
-
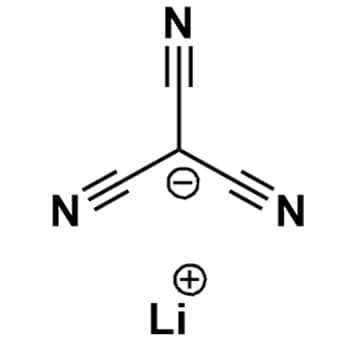
Lithium tricyanomethanide, >99%
$413.22 – $4,848.63Price range: $413.22 through $4,848.63Product Code: KI-0056-HP -
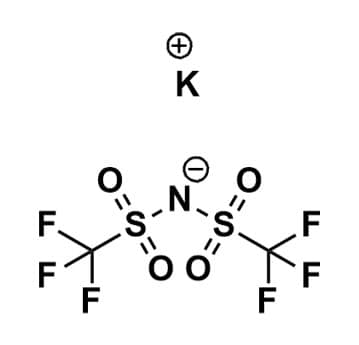
Potassium bis(trifluoromethanesulfonyl)imide, 99%
$49.00 – $377.40Price range: $49.00 through $377.40Product Code: ROCO-011 -
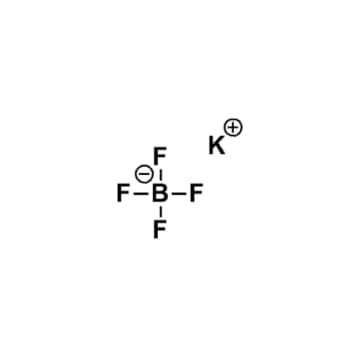
Potassium tetrafluoroborate, >98%
$29.66 – $242.03Price range: $29.66 through $242.03Product Code: KI-0042-HP -
Zinc bis(trifluoromethylsulfonyl)imide, >98%
$120.27 – $1,195.17Price range: $120.27 through $1,195.17Product Code: KI-0062-HP