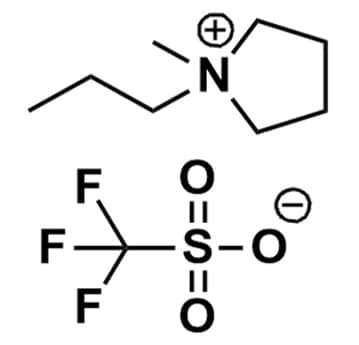
1-Methyl-1-propylpyrrolidinium triflate, >98%
Price range: $309.88 through $3,459.35
Product Code: IL-0162-HPCAS NO: 1224852-54-3
- Chemical Formula: C8H18F3NO3S
- Synonyms: 1-Methyl-1-propylpyrrolidinium trifluoromethanesulfonate, 1-Methyl-1-propylpyrrolidinium trifluoromethanesulfonate, PMPyrr OTf, PYR13 OTf, PY13 OTf
SUM Formula: C9H18F3NO3S
Molecular Weight: 277.31
Melting Point: 76 °C
Purity: >98%
- SUM Formula: C9H18F3NO3S
- Molecular Weight: 277.31
- Melting Point: 76 °C
1-Methyl-1-propylpyrrolidinium triflate, CAS: 1224852-54-3
Key Applications:
Electrolytes for Energy Storage and Conversion
- Serves as a high‑conductivity electrolyte component in lithium, sodium, and multivalent battery systems.
- Enhances electrochemical stability in supercapacitors and hybrid capacitors.
- Used in redox‑active systems where low volatility and high thermal stability are required.
Catalysis and Organic Synthesis
- Functions as a non‑volatile reaction medium for acid‑catalyzed transformations, including alkylation, esterification, and rearrangement reactions.
- Supports transition‑metal catalysis by stabilizing reactive intermediates and improving catalyst lifetime.
- Employed in biphasic and continuous‑flow systems due to its tunable solvation properties.
Separation Science and Extraction
- Applied in liquid–liquid extraction of metal ions, organics, and biomass‑derived compounds.
- Useful in selective extraction processes where hydrophobicity and strong anion coordination improve partitioning efficiency.
- Supports chromatographic method development as a mobile‑phase additive or stationary‑phase modifier.
Polymer and Materials Processing
- Acts as a plasticizer or processing aid in specialty polymers, ionomers, and conductive composites.
- Enables ion transport in polymer electrolytes and gel matrices.
- Used in templating and nanostructuring processes for advanced materials.
Spectroscopy, Electroanalysis, and Fundamental Studies
- Provides a stable, low‑volatility medium for mechanistic electrochemical studies.
- Utilized in spectroscopic investigations of ion pairing, solvation, and charge‑transfer processes.
- Supports development of reference systems and calibration standards in analytical chemistry.
Please contact us if you want to learn more or need assistance with your order.