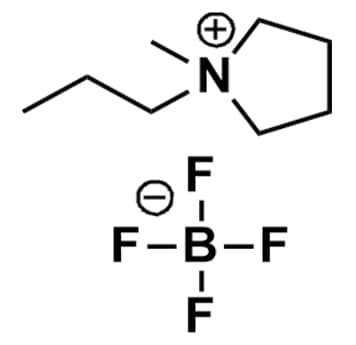
1-Methyl-1-propylpyrrolidinium tetrafluoroborate, >98%
Price range: $240.54 through $10,672.50
Product Code: IL-0147-HPCAS NO: 327022-59-3
- Chemical Formula: C8H18BF4N
- Synonyms: PMPyrr BF4, PYR13 BF4, PY132 BF4, N-Methyl-N-propylpyrrolidinium tetrafluoroborate
- Anion decomposes slowly in the presence of water.
- Weakly-coordinating anion
- Hydrophobic
SUM Formula: C8H18BF4N
Molecular Weight: 215.04
Melting Point: 55 °C
Purity: >98%
- SUM Formula: C8H18BF4N
- Molecular Weight: 215.04
- Melting Point: 55 °C
1-Methyl-1-propylpyrrolidinium tetrafluoroborate, CAS: 327022-59-3
Key Applications:
Electrochemical Systems
- Employed as a non‑volatile electrolyte or electrolyte additive in supercapacitors, lithium‑free metal batteries, and hybrid ion devices.
- Supports wide electrochemical windows, enabling high‑voltage cell designs and improved cycling stability.
- Used in electrodeposition studies for metals and semiconductors where moisture‑sensitive, inert media are required.
Catalysis & Organic Synthesis
- Serves as a tunable reaction medium for transition‑metal catalysis, C–C coupling, oxidation, and hydrogenation reactions.
- Enhances selectivity and yield in ionic‑liquid‑assisted synthesis due to its thermal stability and low coordinating character.
- Utilized in biphasic catalytic systems to improve catalyst recovery and recyclability.
Separation Science & Extraction
- Applied in liquid–liquid extraction of metal ions, dyes, and organic compounds, leveraging its hydrophobicity and adjustable solvation behavior.
- Used in supported ionic liquid membranes (SILMs) for gas separation, CO₂ capture, and pervaporation processes.
Materials Science & Nanotechnology
- Acts as a templating or stabilizing medium for nanoparticle synthesis, enabling controlled morphology and dispersion.
- Incorporated into polymer electrolytes and ionogels to enhance ionic transport and mechanical stability.
- Used in surface modification and thin‑film fabrication where ionic‑liquid‑based processing improves uniformity and adhesion.
Thermal & Chemical Stability Applications
- Suitable for high‑temperature reaction environments due to its low vapor pressure and resistance to decomposition.
- Utilized in studies of ionic‑liquid structure–property relationships, solvation dynamics, and ion transport mechanisms.
Please contact us if you want to learn more or need assistance with your order.