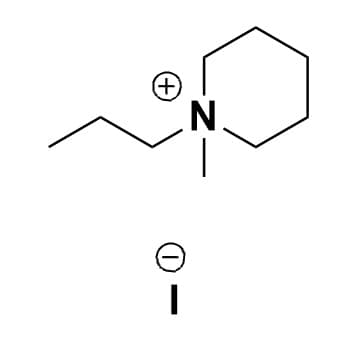
1-Methyl-1-propylpiperidinium iodide, >98%
Price range: $274.53 through $3,085.68
Product Code: IL-0170-HPCAS NO: 17874-63-4
- Chemical Formula: C9H20IN
- Synonyms: N-Methyl-N-propylpiperidinium iodide, PMPip I, PIP13 I
SUM Formula: C9H20IN
Molecular Weight: 269.17
Melting Point: 190 °C
Purity: >98%
- SUM Formula: C9H20IN
- Molecular Weight: 269.17
- Melting Point: 190 °C
1-Methyl-1-propylpiperidinium iodide, CAS: 17874-63-4
Key Applications:
Electrochemical and Energy‑Related Uses
- Redox‑active electrolyte component in halide‑based electrochemical systems, including early‑stage battery and capacitor research where iodide ions participate in charge‑transfer processes.
- Ionic conductivity modifier in prototype electrolytes for dye‑sensitized solar cells (DSSCs) and photoelectrochemical devices that rely on I⁻/I₃⁻ redox couples.
- Supporting electrolyte in voltammetry and electroanalytical studies requiring stable, non‑volatile cations paired with iodide.
Materials Science and Ion‑Transport Studies
- Model compound for ion‑pairing and solvation studies, especially in polar aprotic solvents.
- Precursor for ion‑exchange reactions, enabling synthesis of alternative piperidinium salts (e.g., PF₆⁻, TFSI⁻, BF₄⁻) through metathesis.
- Structure–property investigations in quaternary ammonium systems, including melting point tuning, viscosity behavior, and phase‑transition mapping.
Catalysis and Organic Synthesis
- Phase‑transfer catalyst (PTC) in reactions where iodide enhances nucleophilicity or facilitates halide exchange.
- Halide source for substitution reactions or for generating reactive iodide species in situ.
- Quaternary ammonium scaffold for preparing functionalized piperidinium derivatives used in catalysis or polymer modification.
Polymer and Formulation Research
- Additive in polymer electrolytes to adjust ionic mobility or plasticization behavior.
- Template or structure‑directing agent in the formation of charged polymer networks or ion‑conducting membranes.
- Stabilizer in colloidal or micellar systems where quaternary ammonium cations influence aggregation.
Analytical and Spectroscopic Applications
- Calibration or reference material for NMR, conductivity, or ion‑mobility studies involving quaternary ammonium ions.
- Probe molecule for studying halide–cation interactions, solvation shells, and ion clustering.
Please contact us if you want to learn more or need assistance with your order.