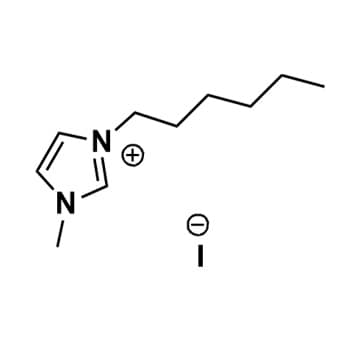
1-Hexyl-2,3-dimethylimidazolium iodide, >98%
Price range: $206.54 through $9,033.43
Product Code: IL-0138-HPCAS NO: 288627-94-1
- Chemical Formula: C11H21IN2
- Synonyms: HDiMIM I, C1C1C6Im I, Im116 I
- Light sensitive
- Aromatic
SUM Formula: C11H21IN2
Molecular Weight: 308.07
Melting Point: 79 °C
Density: 0.666 g/cm³ (25 °C)
Purity: >98%
- SUM Formula: C11H21IN2
- Molecular Weight: 308.07
- Melting Point: 79 °C
- Density: 0.666 g/cm³ (25 °C)
1-Hexyl-2,3-dimethylimidazolium iodide, CAS: 288627-94-1
Key Applications
Solvent and Reaction Medium
- Functions as a hydrophobic ionic liquid suitable for biphasic and phase‑transfer–driven transformations.
- Supports nucleophilic substitution, alkylation, and metal‑catalyzed reactions where iodide enhances substrate activation.
- Useful in moisture‑sensitive or low‑volatility process environments due to its thermal stability and negligible vapor pressure.
Electrochemical and Redox Systems
- Serves as an iodide‑based electrolyte component for redox‑active systems, including dye‑sensitized solar cells (DSSCs) and photoelectrochemical devices.
- Enhances charge transport in systems requiring high iodide concentration and stable cation–anion pairing.
- Suitable for electrodeposition baths where iodide improves metal‑ion complexation and deposition uniformity.
Materials Processing and Thin‑Film Fabrication
- Acts as a templating or structure‑directing ionic liquid for polymeric, inorganic, or hybrid materials.
- Supports controlled crystallization and morphology tuning in halide‑sensitive materials.
- Useful in perovskite precursor formulations where iodide availability influences film quality and defect passivation.
Catalysis and Phase‑Transfer Enhancement
- Provides iodide as a strong nucleophile for catalytic cycles requiring halide exchange or activation.
- Facilitates organocatalytic and metal‑mediated transformations by stabilizing reactive intermediates.
- Effective in biphasic catalysis where the long C6 chain improves partitioning and mass transfer.
Extraction, Separation, and Partitioning
- Enables selective extraction of metal ions, organohalides, and iodine‑containing species through tunable hydrophobicity.
- Supports liquid–liquid extraction systems where iodide enhances complex formation or redox‑driven separations.
- Useful in analytical workflows requiring halide‑rich ionic phases.
Antisolvent and Crystallization Control
- Provides a controlled ionic environment for crystallizing organic salts, pharmaceuticals, and coordination compounds.
- The iodide anion can modulate polymorph formation and solubility behavior in halide‑sensitive systems.
Polymer and Composite Formulations
- Functions as a plasticizing or conductivity‑enhancing additive in ion‑conducting polymers.
- Supports the formation of ionogels and hybrid electrolytes where iodide mobility is required.
Please contact us if you want to learn more or need assistance with your order.