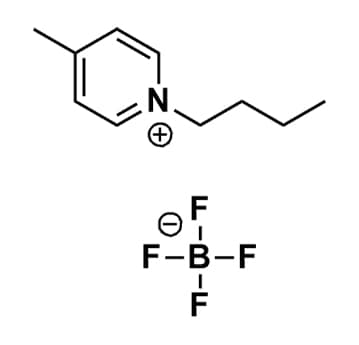
1-Butyl-4-methylpyridinium tetrafluoroborate, >99%
Price range: $190.97 through $8,120.44
Product Code: IL-0085-HPCAS NO: 34952-33-0
- Chemical Formula: C10H16BF4N
- Synonyms: Bu-4-Pic BF4, Bu4Pic BF4, 1-Butyl-4-picolinium tetrafluoroborate, N-Butyl-4-methylpyridinium tetrafluoroborate, N-Butyl-4-picolinium tetrafluoroborate
- Anion decomposes slowly in the presence of water
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 3.18 mS/cm (30 °C)
SUM Formula: C10H16BF4N
Molecular Weight: 237.05
Melting Point: -28 °C
Density: 1.18 g/cm³ (22 °C)
Purity: >99%
Viscosity: 342 cP (20 °C)
- SUM Formula: C10H16BF4N
- Molecular Weight: 237.05
- Melting Point: -28 °C
- Density: 1.18 g/cm³ (22 °C)
- Viscosity: 342 cP (20 °C)
1-Butyl-4-methylpyridinium tetrafluoroborate, CAS: 34952-33-0
Key Applications:
Electrochemical Systems
- Serves as a stable, non‑volatile electrolyte component for supercapacitors and advanced battery chemistries.
- Enhances ionic conductivity and electrochemical window in redox‑active systems.
- Used in electrodeposition studies where controlled ion mobility and low vapor pressure are required.
Catalysis & Organic Synthesis
- Functions as a tunable reaction medium for acid‑ and base‑catalyzed transformations.
- Supports transition‑metal catalysis by improving catalyst dispersion and enabling biphasic separation.
- Applied in alkylation, oxidation, and coupling reactions where ionic liquids improve selectivity and yield.
Separation Science & Extraction
- Employed as a task‑specific solvent for liquid–liquid extraction of metal ions, dyes, and organic contaminants.
- Useful in biphasic catalytic systems and solvent‑swap processes due to its thermal stability and low miscibility with non‑polar phases.
- Supports chromatographic method development as a mobile‑phase additive to modulate retention and peak shape.
Materials Science & Polymer Processing
- Acts as a plasticizing and structuring agent in polymer electrolytes and ion‑conducting films.
- Used to tune morphology and ionic transport in block copolymers and hybrid organic–inorganic materials.
- Facilitates nanoparticle dispersion and surface modification due to its ionic environment and low volatility.
Thermal & Transport Property Studies
- Serves as a model ionic liquid for investigating viscosity, conductivity, and ion‑pairing behavior.
- Used in simulation‑validated experimental studies on solvation dynamics and ion transport mechanisms.
Green Chemistry & Process Intensification
- Enables solvent reduction and recyclability in multi‑step synthetic workflows.
- Supports greener reaction conditions by replacing volatile organic solvents in bench‑scale and pilot‑scale processes.
Please contact us if you want to learn more or need assistance with your order.