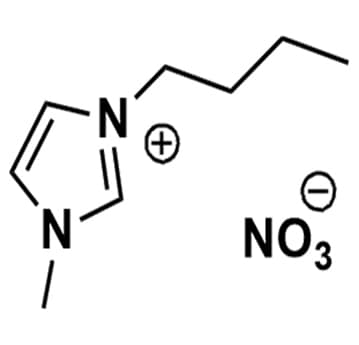
1-Butyl-3-methylimidazolium nitrate, >98%
Price range: $260.45 through $4,022.22
Product Code: IL-0201-HPCAS NO: 179075-88-8
- Chemical Formula: C8H15N3O3
- Synonyms: BMIM NO3 , C 1 C 4 Im NO3 , Im 14 NO3
- Hydrophilic
- Aromatic
*This product will incur a $97.00 HazMat fee when the order is placed.*
Conductivity: 2.59 mS/cm
SUM Formula: C8H15N3O3
Molecular Weight: 201.22
Melting Point: 35°C
Density: 1.16 g/cm³ (25 °C)
Purity: >98%
Viscosity: 136.2 cP
- SUM Formula: C8H15N3O3
- Molecular Weight: 201.22
- Melting Point: 35°C
- Density: 1.16 g/cm³ (25 °C)
- Viscosity: 136.2 cP
1-Butyl-3-methylimidazolium nitrate, CAS: 179075-88-8
Key Applications:
Catalysis & Organic Synthesis
- Polar, non‑volatile reaction medium for nitration, oxidation, and coupling reactions.
- Enhances selectivity and yield in metal‑catalyzed systems.
- Supports biocatalytic transformations by improving enzyme stability and substrate solubility.
Electrochemistry & Energy Materials
- Used as an electrolyte or electrolyte additive in redox systems requiring nitrate‑based ionic conductivity.
- Provides stable ion transport due to its wide liquidus range and thermal stability.
Biomass Processing & Green Chemistry
- Effective solvent for cellulose swelling, lignin dissolution, and biomass fractionation.
- Enables ionic‑liquid‑assisted extraction of bioactive compounds from plant materials.
Materials Science & Nanomaterials
- Templating medium for synthesizing metal nanoparticles, metal oxides, and carbon nanostructures.
- Supports polymer modification and ion‑gel formation to enhance ionic conductivity and mechanical flexibility.
Separation Science & Extraction
- Applied in liquid–liquid extraction of metal ions, including rare earth and transition metals.
- Facilitates selective extraction of aromatic and nitrogen‑containing organic compounds.
Thermal & Propellant Research
- Investigated in energetic material formulations where nitrate anions contribute to controlled oxidizing behavior.
- Used in studies of thermal decomposition pathways relevant to propellant chemistry and combustion modeling.
Please contact us if you want to learn more or need assistance with your order.