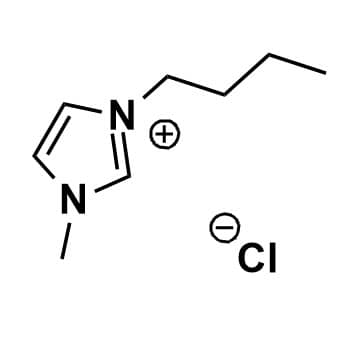
1-Butyl-3-methylimidazolium chloride, >99%
Price range: $80.72 through $1,845.75
Product Code: IL-0014-HPCAS NO: 79917-90-1
- Chemical Fr: C8H15ClN2
- Synonyms: BMIM Cl, C1C4Im Cl
- Hydrophilic
- Aromatic
SUM Formula: C8H15ClN2
Molecular Weight: 174.67
Melting Point: 65 °C
Density: 1.075 g/cm³ (26 °C)
Purity: >99%
- SUM Formula: C8H15ClN2
- Molecular Weight: 174.67
- Melting Point: 65 °C
- Density: 1.075 g/cm³ (26 °C)
1-Butyl-3-methylimidazolium chloride, CAS: 79917-90-1
Key Applications:
1. Biomass Dissolution and Processing
- Highly effective solvent for cellulose, hemicellulose, and lignin, enabling homogeneous dissolution and derivatization.
- Supports biomass fractionation workflows for biofuel, biopolymer, and platform‑chemical production.
- Facilitates pretreatment steps that improve enzymatic hydrolysis efficiency.
2. Desulfurization of Liquid Fuels
- Applied in extractive desulfurization of diesel, gasoline, and other hydrocarbon streams.
- Selectively removes thiophenic and refractory sulfur species under mild conditions.
- Compatible with recyclable extraction cycles, reducing solvent loss and process waste.
3. Electrochemistry and Electroanalytical Studies
- High‑conductivity medium for voltammetry, impedance spectroscopy, and redox‑mechanistic investigations.
- Stabilizes reactive intermediates and enhances signal resolution in electroanalytical measurements.
- Used to study electrode kinetics and ion‑transport behavior in ionic‑liquid environments.
4. Electrodeposition and Materials Fabrication
- Electrolyte component for controlled electrodeposition of metals, alloys, and semiconductors.
- Enables uniform, defect‑minimized coatings due to wide electrochemical stability and low volatility.
- Supports templated nanostructure growth and high‑purity metal film formation.
5. Corrosion Inhibition
- Functions as a corrosion‑inhibiting additive for steel, copper, and aluminum systems.
- Forms protective interfacial layers that suppress anodic and cathodic reactions.
- Used in ionic‑liquid‑based inhibitor formulations for harsh or high‑temperature environments.
6. Extraction and Separation Science
- Effective extractant for metal ions, dyes, sulfur compounds, and polar organics.
- Applied in liquid–liquid extraction, supported ionic‑liquid membranes, and selective partitioning systems.
- Enhances separation efficiency due to strong hydrogen‑bonding and tunable polarity.
7. Catalysis and Organic Synthesis
- Reaction medium for acid‑ and base‑catalyzed transformations, including alkylation, cyclization, and condensation.
- Improves catalyst stability and selectivity in homogeneous and biphasic systems.
- Supports recyclable catalytic phases for greener process design.
8. Polymer Science and Advanced Materials
- Solvent for polysaccharides, polyacrylonitrile, and other high‑polarity polymers.
- Enables homogeneous polymer modification and composite fabrication.
- Used in ion‑conductive polymer electrolytes and membrane development.
9. Nanomaterials and Colloid Stabilization
- Medium for synthesizing metal, oxide, and semiconductor nanoparticles with controlled morphology.
- Acts as a stabilizer and structure‑directing agent in templated nanomaterial growth.
- Facilitates dispersion of carbon nanotubes, graphene, and other nanocarbons.
10. Spectroscopy and Analytical Chemistry
- Solvent for NMR, IR, and UV‑Vis studies of solute–solvent interactions.
- Stabilizes reactive species during mechanistic investigations.
- Used in sample preparation workflows for polar and ionic analytes.
Please contact us if you want to learn more or need assistance with your order.