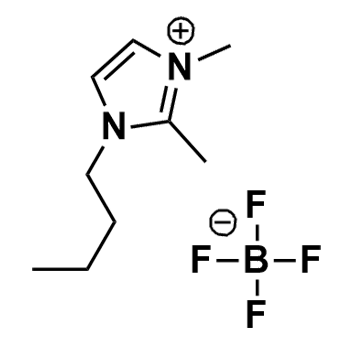
1-Butyl-2,3-dimethylimidazolium tetrafluoroborate, >99%
Price range: $148.58 through $9,179.29
Product Code: IL-0058-HPCAS NO: 402846-78-0
- Chemical Formula: C9H17BF4N2
- Synonyms: BDiMIM BF4, Im114 BF4
- Anion decomposes slowly in the presence of water
- Aromatic
Conductivity: 2.16 mS/cm (45 °C)
SUM Formula: C9H17BF4N2
Molecular Weight: 240.05
Melting Point: 36 °C
Density: 1.239 g/cm³
Purity: >99%
Viscosity: 172 cP (45 °C)
- SUM Formula: C9H17BF4N2
- Molecular Weight: 240.05
- Melting Point: 36 °C
- Density: 1.239 g/cm³
- Viscosity: 172 cP (45 °C)
1-Butyl-2,3-dimethylimidazolium tetrafluoroborate, CAS: 402846-78-0
Key Applications:
Electrochemical Systems
- Serves as a stable, low‑volatility ionic medium for electrochemical studies requiring wide electrochemical windows.
- Supports high ionic conductivity in prototype electrolytes for capacitors, redox systems, and electrodeposition research.
- Useful in evaluating ion‑transport behavior in mixed‑solvent or solvent‑free electrolyte formulations.
Catalysis and Synthesis
- Functions as a non‑volatile reaction medium for acid‑sensitive or moisture‑sensitive transformations where BF₄⁻ stability is advantageous.
- Enables selective catalytic processes involving transition‑metal complexes, particularly where imidazolium cations enhance solubility or ligand behavior.
- Applied in biphasic and recyclable catalytic systems due to its thermal stability and low vapor pressure.
Separation Science
- Employed as a tunable solvent for liquid–liquid extraction of metal ions, organic compounds, and hydrophobic analytes.
- Supports chromatographic method development as an additive or stationary‑phase modifier to adjust polarity and retention behavior.
Materials and Polymer Science
- Acts as a structuring agent or ionic plasticizer in polymer matrices, improving conductivity and mechanical flexibility.
- Used in preparing ionogels and hybrid materials where BF₄⁻ contributes to stable, low‑moisture ionic environments.
- Facilitates dispersion and stabilization of nanoparticles in ionic‑liquid‑based media.
Thermal and Solvation Applications
- Suitable for high‑temperature reaction environments due to its thermal robustness and low flammability.
- Provides a controlled solvation environment for studying solute–ion interactions, viscosity effects, and ionic‑liquid microstructures.
Analytical and Spectroscopic Use
- Serves as a reference ionic medium for NMR, IR, and Raman studies examining cation–anion interactions and solvation dynamics.
- Useful in calibrating or benchmarking physicochemical property measurements such as conductivity, density, and viscosity.
Please contact us if you want to learn more or need assistance with your order.