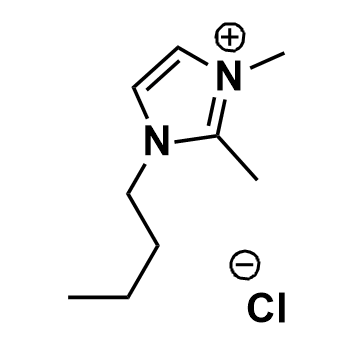
1-Butyl-2,3-dimethylimidazolium chloride, >99%
Price range: $137.20 through $5,851.45
Product Code: IL-0056-HPCAS NO: 98892-75-2
- Chemical Formula: C9H17ClN2
- Synonyms: BDiMIM Cl, Im114 cl
- Hydrophobic
- Aromatic
SUM Formula: C9H17ClN2
Molecular Weight: 188.70
Melting Point: 100 °C
Density: 1.259 g/cm³
Purity: >99%
- SUM Formula: C9H17ClN2
- Molecular Weight: 188.70
- Melting Point: 100 °C
- Density: 1.259 g/cm³
1-Butyl-2,3-dimethylimidazolium chloride, CAS: 98892-75-2
Key Applications:
Solvent and Reaction Medium
- Functions as a stable, low‑volatility ionic liquid for organic synthesis where enhanced cation steric bulk is beneficial.
- Supports transition‑metal‑catalyzed transformations, including cross‑coupling and hydrogenation, by providing a polar, non‑coordinating environment.
- Useful in acid‑sensitive or moisture‑sensitive systems due to its thermal robustness and low vapor pressure.
Electrochemical and Materials Applications
- Serves as an electrolyte component or ionic additive in electrochemical cells, benefiting from its wide electrochemical window and high ionic conductivity.
- Employed in electrodeposition studies where chloride coordination influences metal nucleation and morphology.
- Acts as a structuring agent in polymer–ionic liquid composites, improving ion transport and mechanical stability.
Extraction and Separation Processes
- Effective in biphasic extraction systems for metal ions, organic molecules, and catalytic species, leveraging its tunable polarity and chloride‑based coordination behavior.
- Supports liquid–liquid extraction workflows in analytical chemistry and process development.
Catalysis and Phase‑Transfer Systems
- Functions as a phase‑transfer medium for reactions requiring chloride availability or enhanced solvation of polar intermediates.
- Can be incorporated into supported ionic liquid phases (SILPs) for heterogeneous catalysis, improving catalyst dispersion and recyclability.
Thermal and Process Engineering Uses
- Suitable as a heat‑transfer or thermal‑stabilizing component in high‑temperature process environments where conventional solvents degrade.
- Utilized in studies of ionic‑liquid structure–property relationships, particularly those involving steric modification of imidazolium cations.
Research and Method Development
- Applied in spectroscopic and mechanistic investigations of ionic‑liquid microenvironments, ion pairing, and solvation dynamics.
- Supports formulation screening in materials science, electrochemistry, and catalysis where chloride‑based ionic liquids serve as benchmarks or tuning agents.
Please contact us if you want to learn more or need assistance with your order.