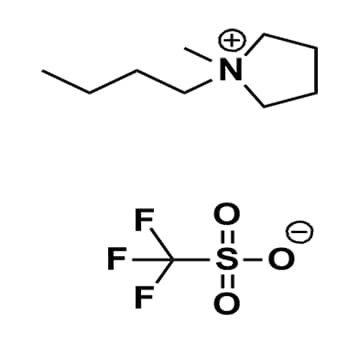
1-Butyl-1-methylpyrrolidinium triflate, >98%
Price range: $171.19 through $2,738.82
Product Code: IL-0113-HPCAS NO: 367522-96-1
- Chemical Formula: C10H20F3NO3S
- Synonyms: BMPyrr OTf, PYR 14 OTf, PY 14 OTf, 1-Butyl-1-methylpyrrolidinium trifluoromethanesulfonate, N-Butyl-N-methylpyrrolidinium trifluoromethanesulfonate
Conductivity: 1.85 mS/cm (24 °C)
SUM Formula: C10H20F3NO3S
Molecular Weight: 291.33
Melting Point: 5 °C
Density: 1.25 g/cm³ (27 °C)
ECW: 4.2V
Purity: >98%
Viscosity: 148 cP (25°C)
- SUM Formula: C10H20F3NO3S
- Molecular Weight: 291.33
- Melting Point: 5 °C
- Density: 1.25 g/cm³ (27 °C)
- ECW: 4.2V
- Viscosity: 148 cP (25°C)
1-Butyl-1-methylpyrrolidinium triflate, CAS: 367522-96-1
Key Applications:
Electrochemistry
- Serves as a stable ionic liquid electrolyte component for systems requiring wide electrochemical windows.
- Supports ion transport in lithium, sodium, and multivalent battery research where low volatility and high thermal stability are essential.
- Used in electrodeposition studies, particularly for metals that benefit from non‑aqueous, low‑moisture environments.
Catalysis and Synthesis
- Functions as a non‑volatile reaction medium for acid‑catalyzed and transition‑metal‑catalyzed transformations.
- Provides a tunable ionic environment that enhances selectivity and reaction rates in organic synthesis.
- Useful in biphasic catalysis where phase separation and recyclability of the ionic liquid are advantageous.
Materials Science
- Incorporated into polymer electrolytes and ionogels to improve ionic conductivity and mechanical stability.
- Applied in the fabrication of membranes for gas separation, proton conduction, and advanced filtration.
- Used in surface modification and thin‑film processing where controlled ionic environments are required.
Analytical and Separation Science
- Acts as an additive in chromatography and electrophoresis to adjust selectivity, peak shape, and ion‑pairing behavior.
- Serves as a solvent or co‑solvent for spectroscopic studies requiring low vapor pressure and high chemical stability.
Thermal and Process Engineering
- Employed as a heat‑transfer or thermal‑storage medium in systems requiring high thermal stability and non‑flammability.
- Used in solvent extraction processes where immiscibility with organic phases and tunable polarity are beneficial.
Please contact us if you would like to learn more or require assistance with your order.