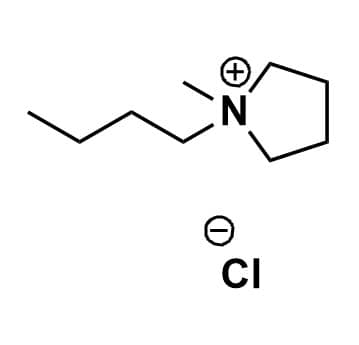
1-Butyl-1-methylpyrrolidinium chloride, >99%
Price range: $117.42 through $3,612.26
Product Code: IL-0075-HPCAS NO: 479500-35-1
- Chemical Formula: C9H20ClN
- Synonyms: BMPyrr Cl, PYR14 Cl, PY14 Cl, N-Butyl-N-methylpyrrolidinium chloride
- Hydrophilic
SUM Formula: C9H20ClN
Molecular Weight: 177.72
Melting Point: 198 °C
Density: 1.219 g/cm³
Purity: >99%
- SPECIFIC GRAVITY: NA
- SUM Formula: C9H20ClN
- Molecular Weight: 177.72
- Melting Point: 198 °C
- Density: 1.219 g/cm³
1-Butyl-1-methylpyrrolidinium Chloride, CAS: 479500-35-1
Key Applications:
Electrochemical Systems & Ionic Liquid Research
- Serves as a precursor for synthesizing pyrrolidinium‑based ionic liquids with tunable viscosity, conductivity, and thermal stability.
- Used in studies evaluating cation structure–property relationships in electrolytes for batteries, supercapacitors, and electrochemical sensors.
- Supports development of chloride‑based molten salt electrolytes for high‑temperature electrochemical processes.
Phase‑Transfer & Catalytic Media
- Functions as a phase‑transfer catalyst in organic transformations requiring efficient ion shuttling between immiscible phases.
- Employed as a reaction medium for nucleophilic substitution, alkylation, and polymerization reactions where low‑volatility ionic environments improve selectivity.
Materials Science & Polymer Processing
- Used to tune ionic conductivity and mechanical properties in polymer electrolytes and ion‑conducting membranes.
- Incorporated into ionogels and hybrid materials to enhance ionic mobility and thermal stability.
- Supports fabrication of advanced materials where chloride counterions enable controlled ion exchange or post‑synthetic modification.
Surfactant, Solvation, & Self‑Assembly Studies
- Applied in micellization and solvation research to probe how pyrrolidinium cations influence aggregation behavior.
- Useful in studying ion–solvent interactions, hydrogen‑bonding networks, and structural organization in ionic liquid mixtures.
Extraction, Separation, & Coordination Chemistry
- Acts as a chloride‑donating ionic medium for metal ion extraction, speciation, and coordination studies.
- Enables selective solubilization of metal salts and organometallic complexes in non‑aqueous systems.
Analytical Chemistry & Sample Preparation
- Utilized as an additive in chromatographic and electrophoretic methods to modify ion‑pairing behavior and improve separation efficiency.
- Supports sample pretreatment workflows requiring stable, non‑volatile ionic matrices.
Please contact us if you want to learn more or need assistance with your order.