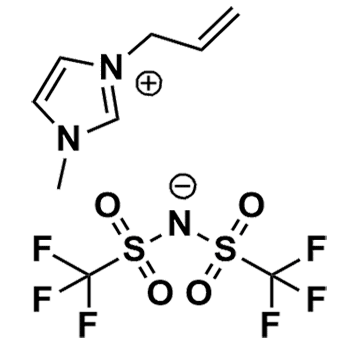
1-Allyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $164.15 through $2,261.81
Product Code: IL-0239-HPCAS NO: 655249-87-9
- Chemical Formula: C9H11F6N3O4S2
- Synonyms: AllylMIM BTA, AllylMIM NTf 2, AllylMIM TFSI, AllylMIM BTI
- Weakly-coordinating anion
- Polymerizable
- Hydrophobic
- Aromatic
Conductivity: 8.871 mS/cm (30 °C)
SUM Formula: C9H11F6N3O4S2
Molecular Weight: 403.32
Melting Point: <25 °C
Density: 1.494 (29°C)
ECW: 4.8V
Purity: >99%
Viscosity: 35 cP (25 °C)
- SUM Formula: C9H11F6N3O4S2
- Molecular Weight: 403.32
- Melting Point: <25 °C
- Density: 1.494 (29°C)
- ECW: 4.8V
- Viscosity: 35 cP (25 °C)
1-Allyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 655249-87-9
Key Applications:
Electrochemical Systems
- Serves as a high‑conductivity ionic medium for supercapacitors, redox‑flow systems, and advanced electrochemical cells.
- Supports wide electrochemical windows, enabling stable operation in high‑voltage environments.
- Useful as a co‑solvent or electrolyte additive to improve ion mobility and reduce interfacial resistance.
Polymer and Materials Processing
- Acts as a reactive ionic liquid for polymer modification, particularly where the allyl group enables covalent incorporation into polymer matrices.
- Facilitates dissolution and processing of polar polymers, including cellulose derivatives and specialty engineering plastics.
- Functions as a plasticizer or ionic dopant in ion‑conducting polymer films.
Catalysis and Organic Synthesis
- Provides a tunable, low‑volatility reaction medium for transition‑metal catalysis, nucleophilic substitutions, and alkylation reactions.
- The allyl substituent can participate in or stabilize reactive intermediates, offering unique selectivity profiles in certain catalytic cycles.
- Enables biphasic catalysis where product separation is simplified due to immiscibility with common organic solvents.
Separation Science and Extraction
- Effective as an extraction phase for metal ions, hydrophobic organics, and fluorinated species due to the hydrophobic TFSI anion.
- Supports task‑specific extraction workflows where the allyl group can be leveraged for further functionalization or immobilization on solid supports.
Surface and Interface Engineering
- Useful in forming ionically conductive surface films for sensors, coatings, and thin‑film devices.
- The allyl moiety allows covalent grafting onto surfaces via radical or photochemical pathways, enabling durable ionic interfaces.
Energy and Photonic Materials
- Employed in dye‑sensitized solar cells and related optoelectronic systems as a stable ionic medium with low volatility.
- Enhances charge transport in composite materials containing conductive polymers or nanostructured carbon.
General Solvent and Processing Uses
- Provides a thermally stable, non‑volatile solvent for high‑temperature reactions and moisture‑sensitive transformations.
- Compatible with a wide range of inorganic salts, enabling formulation of designer electrolytes and ionic mixtures.
Please contact us if you want to learn more or need assistance with your order.