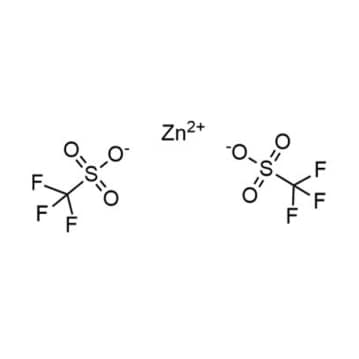
Zinc trifluoromethansulfonate, >99%
Price range: $155.75 through $1,450.67
Product Code: ZBE-0100-HPCAS NO: 54010-75-2
- Chemical Formula: C2F6S2O6Zn
- Synonyms: Bis(trifluoromethanesulfonato)zinc, Zinc trifluoromethylsulfonate, Zn(OTf)2, Trifluoromethanesulfonic acid zinc salt, Zinc triflate, Zn(OTf)2
- Materials for Zn-Air Batteries
SUM Formula: C2F6S2O6Zn
Molecular Weight: 363.53
Purity: >99%
- SUM Formula: C2F6S2O6Zn
- Molecular Weight: 363.53
Zinc trifluoromethansulfonate, CAS: 54010-75-2
Key Applications:
Catalysis & Organic Synthesis
- Highly effective Lewis acid catalyst for Friedel–Crafts acylation/alkylation, Diels–Alder reactions, Mukaiyama aldol reactions, and selective esterifications.
- Promotes C–C, C–O, and C–N bond formation under mild, water‑tolerant conditions.
- Enables solvent-free and green synthetic routes, supporting sustainable process development.
Electrolytes for Energy Storage
- Used as a high‑purity zinc salt in aqueous and non‑aqueous zinc‑ion battery electrolytes, improving ionic conductivity and cycling stability.
- Supports high‑voltage, dendrite‑suppressed electrolyte formulations due to its stable, non‑coordinating triflate anion.
Polymerization & Materials Chemistry
- Functions as a polymerization initiator or co‑catalyst in cationic and ring‑opening polymerizations.
- Enhances crosslinking efficiency in specialty polymers and elastomers.
- Applied in the synthesis of functional materials, including ionomers and fluorinated polymer systems.
Ionic Liquid & Deep Eutectic Solvent (DES) Formulations
- Serves as a metal triflate precursor for designing task‑specific ionic liquids.
- Used in DES systems for metal processing, catalysis, and electrochemical applications.
Coordination Chemistry & Metal–Organic Frameworks (MOFs)
- Provides a clean, non‑interfering triflate counterion for zinc coordination complexes.
- Supports MOF synthesis where controlled zinc coordination and high anion stability are required.
Fine Chemicals, Pharmaceuticals & Specialty Intermediates
- Applied in selective protection/deprotection strategies, acetalization, and glycosylation.
- Useful in stereoselective transformations where mild Lewis acidity is essential.
- Employed in high‑purity synthesis workflows due to its low water content and excellent solubility profile.