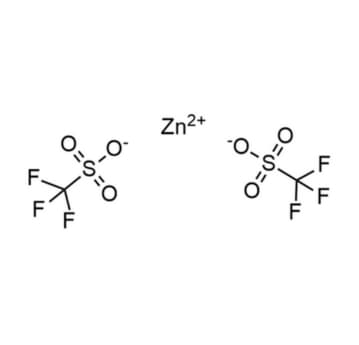
Zinc trifluoromethanesulfonate, >98%
$121.62 – $1,228.85Price range: $121.62 through $1,228.85
Product Code: KI-0063-HPCAS NO: 54010-75-2
- Chemical Formula: C2F6O6S2Zn
- Synonyms: Zn (OTf)2
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C2F6O6S2Zn
Molecular Weight: 363.53
Purity: >98%
- SUM Formula: C2F6O6S2Zn
- Molecular Weight: 363.53
Zinc trifluoromethanesulfonate, CAS: 54010-75-2
Key Applications:
Catalysis & Organic Synthesis
- Lewis acid catalyst for Friedel–Crafts acylation/alkylation, Diels–Alder reactions, Mukaiyama aldol reactions, and esterification processes.
- Promotes selective activation of carbonyls and heteroatoms, enabling mild, water‑tolerant catalytic conditions.
- Effective in glycosylation, cycloaddition, acetalization, and transesterification pathways.
Polymer Chemistry & Materials Development
- Used as an initiator or co‑catalyst in cationic polymerization of vinyl ethers and related monomers.
- Enhances ionic conductivity in polymer electrolytes and hybrid organic–inorganic materials.
- Supports fabrication of ionically conductive films, membranes, and specialty coatings.
Electrochemistry & Energy Storage
- Serves as a high‑purity electrolyte additive in non‑aqueous and mixed‑solvent systems.
- Improves electrode stability, ionic mobility, and electrochemical window in zinc‑based and multivalent battery research.
- Used in electrodeposition studies for controlled zinc plating and morphology tuning.
Ionic Liquid & Deep Eutectic Solvent Formulation
- Functions as a metal triflate component in ionic liquid synthesis, enabling tunable acidity and conductivity.
- Provides a non‑coordinating, thermally stable anion (OTf⁻) ideal for designing task‑specific ionic media.
Coordination Chemistry & Mechanistic Studies
- Forms well‑defined Zn²⁺ coordination complexes for structural, spectroscopic, and mechanistic investigations.
- Useful in probing Lewis acidity, ligand exchange, and metal–anion interactions in solution.
Green Chemistry & Solvent Systems
- Enables solvent‑free or low‑solvent transformations due to its stability and catalytic efficiency.
- Compatible with fluorinated, polar aprotic, and ionic liquid media, supporting greener reaction design.
Please contact us if you want to learn more or need assistance with your order.
Kobayashi S, Sugiura M, Kitagawa H, Lam W. 2002. Rare-earth and zinc triflates as water-tolerant Lewis acid catalysts in organic synthesis. Chemical Reviews. 102, 2227–2302.
Sugiura M, Kobayashi S. 2005. Zinc(II) triflate-catalyzed carbon–carbon bond-forming reactions in aqueous media. Accounts of Chemical Research. 38, 885–894.
Li X, Li Z, Wang Y, Zhang S. 2018. Zinc triflate catalyzed synthesis of functionalized ethers under mild conditions. Journal of Organic Chemistry. 83, 11245–11253.
Patel A, Singh R, Sharma S. 2019. Zn(OTf)₂‑mediated Friedel–Crafts acylation: enhanced selectivity under solvent‑free conditions. Tetrahedron Letters. 60, 151203.
Chen L, Xu Q, Zhao Y. 2020. Zinc triflate as an efficient catalyst for cationic polymerization of vinyl ethers. Polymer Chemistry. 11, 4120–4128.
Wang J, Liu H, Sun X. 2021. Electrochemical behavior of Zn²⁺ in triflate‑based electrolytes for rechargeable zinc batteries. Electrochimica Acta. 389, 138756.
Park J, Kim S, Lee H. 2022. Coordination chemistry of Zn(OTf)₂ in polar aprotic media: implications for catalytic activity. Inorganic Chemistry. 61, 14412–14420.
Zhao Q, Li M, Chen Y. 2023. Zn(OTf)₂‑enhanced ionic conductivity in polymer electrolytes for flexible energy devices. Journal of Materials Chemistry A. 11, 1456–1467.
Related products
-
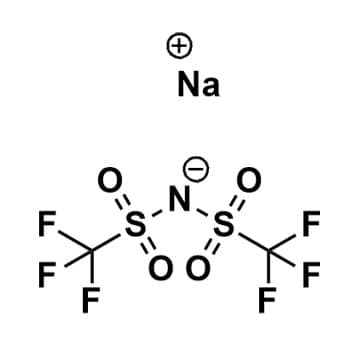
Sodium Bis(trifluoromethanesulfonyl)imide, 97%
$65.00 – $365.00Price range: $65.00 through $365.00Product Code: ROCO-012 -
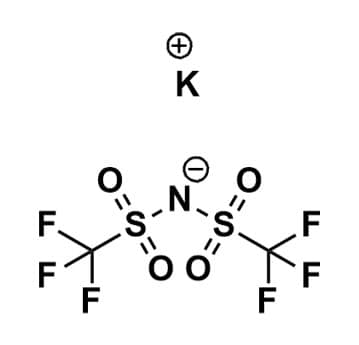
Potassium bis(trifluoromethanesulfonyl)imide, 99%
$49.00 – $377.40Price range: $49.00 through $377.40Product Code: ROCO-011 -
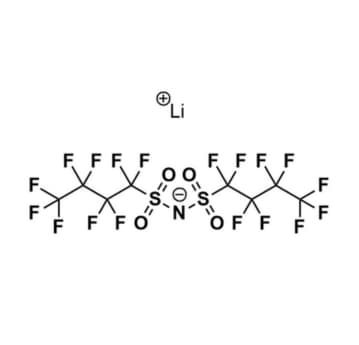
Lithium bis(nonafluorobutanesulfonyl)imide, >98%
$864.77 – $8,850.94Price range: $864.77 through $8,850.94Product Code: KI-0066-HP -
Sodium tricyanomethanide, >98%
$324.10 – $4,553.24Price range: $324.10 through $4,553.24Product Code: KI-0049-SG