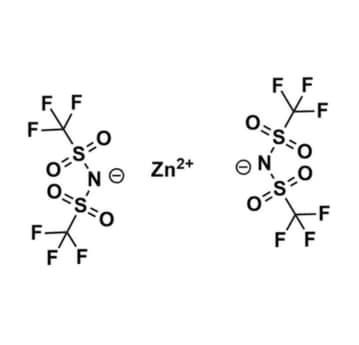
Zinc bis(trifluoromethylsulfonyl)imide, >98%
$120.27 – $1,195.17Price range: $120.27 through $1,195.17
Product Code: KI-0062-HPCAS NO: 168106-25-0
- Chemical Formula: C4F12N2O8S4Zn
- Synonyms: Zn (BTA)2
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C4F12N2O8S4Zn
Molecular Weight: 625.68
Purity: >98%
- SUM Formula: C4F12N2O8S4Zn
- Molecular Weight: 625.68
Zinc bis(trifluoromethylsulfonyl)imide, CAS: 168106-25-0
Key Applications:
Electrolyte development for zinc‑based batteries
Used as a high‑conductivity zinc salt in aqueous, organic, and ionic‑liquid electrolyte systems. The weakly coordinating TFSI⁻ anion supports wide electrochemical stability windows, enhanced ion mobility, and reduced dendrite formation in zinc‑ion and hybrid multivalent batteries.
Supercapacitors and hybrid energy‑storage devices
Serves as a charge carrier in high‑voltage supercapacitor electrolytes, enabling improved cycling stability and tunable ionic transport in organic and ionic‑liquid media.
Electrodeposition and surface engineering
Facilitates controlled zinc electrodeposition with smoother morphology and reduced passivation. Applied in research on corrosion‑resistant coatings, thin‑film growth, and mechanistic studies of metal nucleation and interfacial behavior.
Ionic liquid and deep eutectic solvent formulation
Acts as a metal‑centered component in the synthesis of task‑specific ionic liquids and eutectic systems. The TFSI⁻ counterion enables low viscosity, high conductivity, and broad solvent compatibility for catalysis, separations, and electrochemical media.
Lewis acid catalysis and organic synthesis
Functions as a non‑nucleophilic Lewis acid suitable for activating oxygen‑, nitrogen‑, and sulfur‑containing substrates. Its high solubility in low‑polarity solvents and chemically inert anion minimize side reactions in polymerization and fine‑chemical transformations.
Materials processing and thin‑film research
Used as a precursor or dopant in polymer modification, vapor‑phase deposition studies, and ion‑exchange processes. Supports investigations into dielectric behavior, ion transport, and interfacial stability in soft materials and functional films.
Please contact us if you want to learn more or need assistance with your order.
Related products
-
1-Methyl-1-propylpyrrolidinium bis(trifluoromethylsulfonyl)imide, >99.5%
$106.05 – $1,379.98Price range: $106.05 through $1,379.98Product Code: IL-0044-UP -
1-Butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, >99.5%
$106.18 – $5,520.31Price range: $106.18 through $5,520.31Product Code: IL-0029-UP -
1-Ethyl-3-methylimidazolium bis(fluorosulfonyl)imide, >99%
$91.96 – $1,224.36Price range: $91.96 through $1,224.36Product Code: IL-0342-HP -
1-Methyl-3-tetradecylimidazolium chloride, >98%
$243.38 – $2,855.90Price range: $243.38 through $2,855.90Product Code: IL-0141-HP