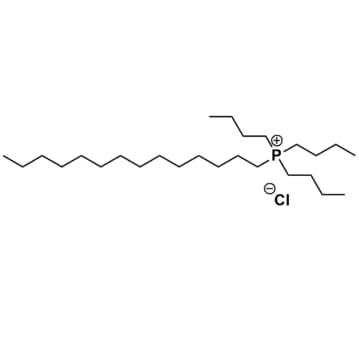
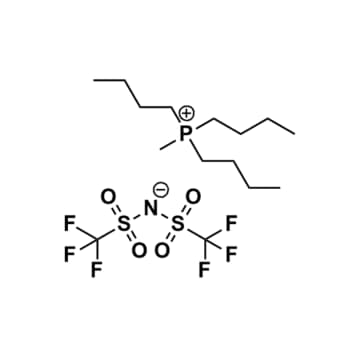
Tributyltetradecylphosphonium chloride, >95%
Price range: $77.74 through $1,744.79
Product Code: IN-0017-TGCAS NO: 81741-28-8
- Chemical Formula: C26H55ClP
- Synonyms: P444(14) Cl
** This product will incur a $97.00 HazMat fee when the order is placed. **
SUM Formula: C26H55ClP
Molecular Weight: 435.15
Melting Point: 42-43 °C
Purity: >95%
- SUM Formula: C26H55ClP
- Molecular Weight: 435.15
- Melting Point: 42-43 °C
Tributyltetradecylphosphonium chloride, CAS: 81741-28-8
Key Applications:
Phase‑Transfer & Solvent‑Free Catalysis
- Functions as a highly efficient phase‑transfer catalyst for nucleophilic substitutions, alkylations, and condensations in biphasic or solvent‑minimized systems.
- Enhances reaction rates in ionic‑liquid‑like media, enabling greener processing and improved mass transfer.
Surfactant, Emulsifier & Hydrotrope
- Long‑chain phosphonium cation provides strong surface activity, supporting emulsification, dispersion, and wetting in complex formulations.
- Used in oil–water systems, specialty cleaners, and industrial processing aids requiring thermal and chemical robustness.
Antimicrobial & Biocidal Formulations
- Exhibits broad‑spectrum antimicrobial activity, making it suitable for research into disinfectants, coatings, and preservation systems.
- Evaluated in polymeric matrices and surface treatments where durable cationic charge is beneficial.
Polymer Modification & Compatibilization
- Acts as a cationic additive to tune polymer polarity, conductivity, and interfacial behavior.
- Supports dispersion of fillers, nanoparticles, and pigments in hydrophobic matrices.
Electrochemical & Ionic‑Liquid Research
- Serves as a precursor or component in task‑specific ionic liquids, especially those requiring high hydrophobicity and thermal stability.
- Used in studies of ion transport, electrode interfaces, and solvent‑free electrochemical systems.
Extraction, Separation & Phase Behavior Studies
- Effective in liquid–liquid extraction of metal ions and organics due to strong ion‑pairing and hydrophobicity.
- Supports research into solvent structuring, micelle formation, and amphiphilic ionic systems.
You may also like…
-
1-Ethyl-3-methylimidazolium chloride, >98%
Price range: $118.78 through $5,050.34Product Code: IL-0093-HP -
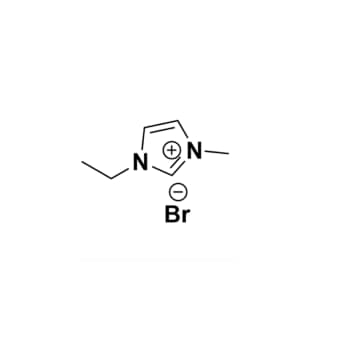
1-Ethyl-3-methylimidazolium bromide, >99%
Price range: $104.69 through $2,471.33Product Code: IL-0015-HP -
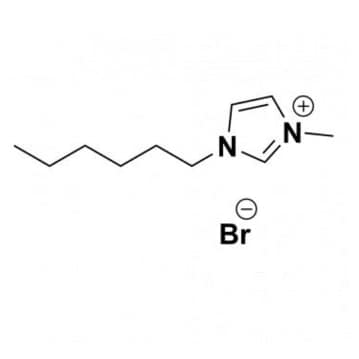
1-Hexyl-3-Methylimidazolium Bromide, >99%
Price range: $161.31 through $1,039.20Product Code: IL-0069-HP -
1-Butyl-1-methylpiperidinium bromide, >99%
Price range: $151.42 through $3,466.39Product Code: IL-0153-HP