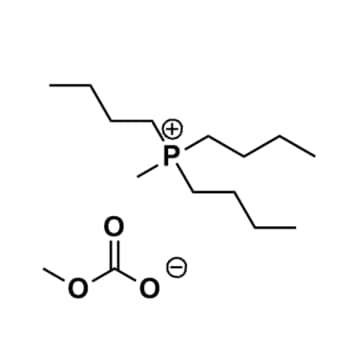
Tributylmethylphosphonium methylcarbonate, >97%, 50% in MeOH
Price range: $193.81 through $1,821.24
Product Code: IN-0030-SGCAS NO: 120256-45-3
- Chemical Formula: C15H33O3P
- Synonyms: P1444 BTA, P1444 NTf2, P1444 TFSI, P1444 BTI
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C15H33O3P
Molecular Weight: 292.22
Purity: >97%
- SUM Formula: C15H33O3P
- Molecular Weight: 292.22
Tributylmethylphosphonium methylcarbonate, CAS: 120256-45-3
Key Applications:
CO₂‑Derived Chemistry & Green Carbonylation
- Functions as a reactive methylcarbonate donor in carbonylation, carboxymethylation, and carbonate‑forming reactions.
- Supports CO₂‑utilization pathways, particularly in systems where methylcarbonate anions act as masked CO₂ equivalents.
- Useful in developing sustainable synthesis routes for carbonates, carbamates, and alkyl carbonates.
Phase‑Transfer & Catalytic Media
- Acts as an efficient phase‑transfer medium for nucleophilic substitution, esterification, and alkylation reactions.
- Enhances reaction rates in biphasic and low‑polarity systems due to its high polarity and low volatility.
- Serves as a tunable solvent/catalyst hybrid in organophosphonium‑mediated transformations.
Polymer & Materials Processing
- Employed as a processing aid and solvation medium for engineering polymers, including polycarbonates, polyurethanes, and cellulose derivatives.
- Supports polymer functionalization via carbonate exchange or mild alkylation pathways.
- Useful in membrane casting, ion‑conductive films, and surface modification workflows.
Electrochemical & Energy Applications
- Provides a stable ionic environment for electrolyte formulation, especially where carbonate anions improve SEI formation or interfacial stability.
- Investigated in CO₂‑electroreduction, capacitive systems, and low‑volatility electrolytes requiring wide electrochemical windows.
Extraction, Separation & Solvent Systems
- Effective in liquid–liquid extraction of metal ions, organics, and CO₂‑reactive species.
- Offers tunable polarity for selective separations, particularly where carbonate coordination or mild basicity is advantageous.
- Useful as a green solvent alternative in replacing volatile organic carbonates.
Specialty Synthesis & Fine Chemicals
- Enables selective O‑methylation, carbonate exchange, and mild base‑promoted transformations.
- Serves as a reagent/medium for preparing functionalized carbonates, protected alcohols, and intermediate building blocks.
Please contact us if you want to learn more or need assistance with your order.
You may also like…
-
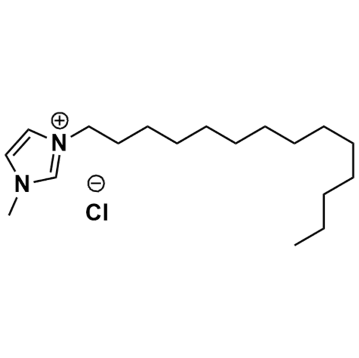
1-Ethyl-3-methylimidazolium chloride, >98%
Price range: $118.78 through $5,050.34Product Code: IL-0093-HP -
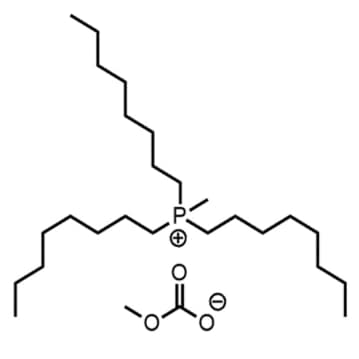
Methyltrioctylphosphonium methylcarbonate, >97%, 30% in MeOH
Price range: $202.34 through $515.79Product Code: IN-0031-SG -
Tributylmethylphosphonium methylcarbonate, >97%, 50% in MeOH
Price range: $193.81 through $1,821.24Product Code: IN-0030-SG