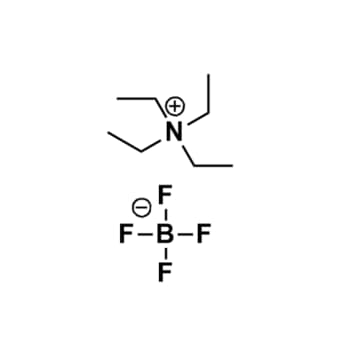
Tetraethylammonium Tetrafluoroborate, >98%
Price range: $124.47 through $774.16
Product Code: IL-0348-HPCAS NO: 429-06-1
- Chemical Formula: C8H20BF4N
- Synonyms: N2222 BF4
For Ultra-Purity of >99%, refer to IL-0348-UP
SUM Formula: C8H20BF4
Molecular Weight: 217.06
Melting Point: >300 °C
Density: 0.996 g/cm³
Purity: >98%
- SUM Formula: C8H20BF4
- Molecular Weight: 217.06
- Melting Point: >300 °C
- Density: 0.996 g/cm³
Tetraethylammonium Tetrafluoroborate, CAS: 429-06-1
Key Applications:
Electrochemical Systems
- Serves as a supporting electrolyte in non‑aqueous electrochemical studies, providing high ionic conductivity and wide electrochemical stability.
- Used in cyclic voltammetry, chronoamperometry, and electrode kinetics research where a non‑coordinating cation is required.
- Enables stable ion transport in organic solvents such as acetonitrile, propylene carbonate, and DMF.
Battery and Energy Storage Research
- Functions as an electrolyte salt in prototype supercapacitors and experimental battery systems where BF₄⁻ anions offer good oxidative stability.
- Supports investigations into ion‑solvent interactions and transport mechanisms in emerging energy‑storage chemistries.
Ionic Liquid and Molten Salt Systems
- Acts as a precursor or dopant in the formulation of tetrafluoroborate‑based ionic liquids.
- Used to tune viscosity, conductivity, and thermal behavior in IL mixtures for electrochemical and separations research.
Phase‑Transfer and Catalysis
- Employed as a phase‑transfer electrolyte in organic synthesis where quaternary ammonium cations improve solubility and reaction rates.
- Useful in catalytic systems requiring weakly coordinating anions to minimize interference with active metal centers.
Analytical Chemistry
- Applied in ion‑pair chromatography and electrophoretic methods to adjust mobile‑phase ionic strength and selectivity.
- Supports mass spectrometry sample preparation where non‑nucleophilic salts are preferred.
Materials Science
- Incorporated into polymer electrolytes and conductive films to study ion mobility and dielectric behavior.
- Used in templating and self‑assembly studies where quaternary ammonium ions influence structural organization.
Please contact us if you want to learn more or need assistance with your order.