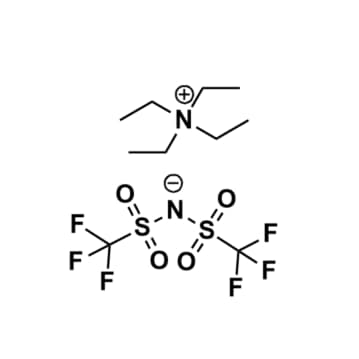
Tetraethylammonium bis(trifluoromethylsulfonyl)imide, >98%
Price range: $124.47 through $6,518.21
Product Code: IL-0335-HPCAS NO: 161401-26-9
- Chemical Formula: C10H20F6N2O4S2
- Synonyms: N2222 BTA, N2222 NTf2, N2222 TFSI, N2222 BTI
- Weakly-coordinating anion
SUM Formula: C10H20F6N2O4S2
Molecular Weight: 410.40
Melting Point: >RT
Purity: >98%
- SUM Formula: C10H20F6N2O4S2
- Molecular Weight: 410.40
- Melting Point: >RT
Tetraethylammonium bis(trifluoromethylsulfonyl)imide, CAS: 161401-26-9
Key Applications:
Electrochemical Systems
- Serves as a supporting electrolyte in non‑aqueous electrochemical studies due to its high ionic conductivity and wide electrochemical stability window.
- Used in voltammetry, electrosynthesis, and redox‑active material characterization where low coordinating anions minimize ion–electrode interactions.
- Enables stable ion transport in aprotic solvents such as acetonitrile, propylene carbonate, and sulfone‑based media.
Battery and Energy Storage Research
- Functions as a model quaternary ammonium cation for studying ion mobility and solvation in next‑generation electrolytes.
- Supports research on high‑voltage electrolyte formulations where TFSI– anions improve oxidative stability.
- Used in fundamental studies of ion pairing, dielectric relaxation, and transport mechanisms relevant to Li‑ion and Na‑ion battery development.
Ionic Liquid and Deep Eutectic Solvent Design
- Acts as a precursor salt for synthesizing tailored ionic liquids via metathesis with lithium, imidazolium, pyrrolidinium, or phosphonium partners.
- Useful for tuning viscosity, polarity, and hydrophobicity in custom IL formulations.
- Supports structure–property investigations of TFSI‑based ionic liquids.
Materials Science and Polymer Electrolytes
- Incorporated into polymer matrices (e.g., PEO, PMMA, PVDF‑HFP) to enhance ionic conductivity and mechanical flexibility.
- Used in studies of ion transport in solid and gel polymer electrolytes.
- Helps evaluate cation–polymer interactions due to the compact, symmetric tetraethylammonium cation.
Spectroscopy and Fundamental Physical Chemistry
- Employed as a benchmark salt for NMR, FTIR, Raman, and dielectric spectroscopy to probe ion pairing and solvation dynamics.
- Supports thermodynamic studies involving ion association, activity coefficients, and transport numbers in non‑aqueous media.
Phase Behavior and Solubility Studies
- Used to map phase diagrams of electrolyte–solvent systems, especially in highly fluorinated or low‑polarity solvents.
- Helps characterize miscibility, ion clustering, and solvent–salt interactions relevant to electrolyte engineering.
Please contact us if you want to learn more or need assistance with your order.