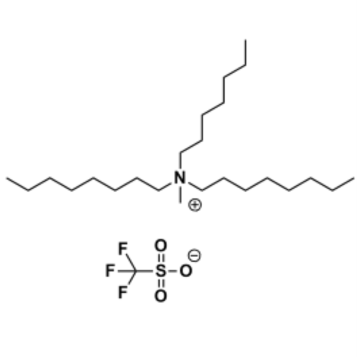
Methyltrioctylammonium triflate, >97%
Price range: $191.10 through $2,498.28
Product Code: IL-0118-HPCAS NO: 121107-18-4
- Chemical Formula: C26H54F3NO3S
- Synonyms: N1888 OTf, Methyltrioctylammonium trifluoromethanesulfonate
- Hydrophobic
SUM Formula: C26H54F3NO3S
Molecular Weight: 517.77
Melting Point: 55–60 °C
Purity: >97%
- SUM Formula: C26H54F3NO3S
- Molecular Weight: 517.77
- Melting Point: 55–60 °C
Methyltrioctylammonium triflate, CAS: 121107-18-4
Key Applications:
Phase‑Transfer and Ion‑Exchange Applications
- Functions as a hydrophobic quaternary ammonium triflate salt for transferring anionic species into nonpolar or moderately polar organic media.
- Supports biphasic and multiphasic reaction systems where strong, non‑coordinating anions are required to stabilize reactive intermediates.
- Useful in ion‑exchange processes where a bulky, lipophilic cation paired with a weakly coordinating anion improves selectivity and extraction efficiency.
Catalysis and Organic Synthesis
- Serves as a non‑nucleophilic ionic component in Lewis and Brønsted acid‑mediated transformations.
- Enhances reaction rates in alkylation, acylation, and rearrangement chemistry by providing a stable triflate counterion that does not interfere with catalytic cycles.
- Employed in systems requiring low water activity and high thermal stability.
Solvent and Electrolyte Engineering
- Acts as a structure‑directing ionic additive in nonaqueous electrolyte formulations.
- Supports high‑conductivity media for electrochemical studies where hydrophobicity and anion stability are critical.
- Useful in tailoring viscosity, polarity, and ion‑pairing behavior in mixed organic solvents.
Extraction and Separation Science
- Facilitates selective extraction of metal ions, particularly in systems where the triflate anion improves solubility of metal complexes in organic phases.
- Applied in liquid–liquid extraction workflows for rare‑earth, transition‑metal, and actinide separations.
- Suitable for designing task‑specific ionic liquids for hydrometallurgical operations.
Materials and Polymer Processing
- Functions as a dopant or ionic plasticizer in specialty polymers, membranes, and ionogels.
- Supports fabrication of hydrophobic ionic matrices with enhanced thermal and chemical stability.
- Useful in templating or modifying surface charge in advanced materials.
Analytical and Spectroscopic Uses
- Provides a stable, non‑coordinating triflate environment for NMR, IR, and MS studies of reactive cationic species.
- Employed as an ion‑pairing reagent in chromatographic method development, especially for hydrophobic analytes.
Please contact us if you want to learn more or need assistance with your order.