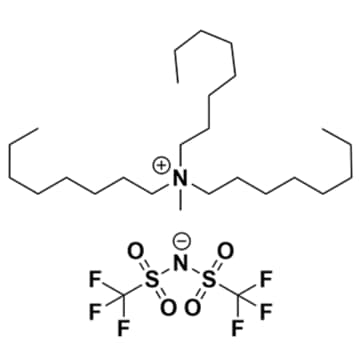
Methyltrioctylammonium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $164.15 through $8,170.01
Product Code: IL-0017-HPCAS NO: 375395-33-8
- Chemical Formula: C12H19F6N3O4S2
- Synonyms: N1888 BTA, N1888 NTf2, N1888 TFSI, N1888 BTI, OMA BTA
- Weakly-coordinating anion
- Hydrophobic
Conductivity: 0.046 mS/cm (20 °C)
SUM Formula: C27H54F6N2O4S2
Molecular Weight: 648.85
Melting Point: -39 °C
Density: 1.15 g/cm³
ECW: 5.1 V
Purity: >99%
Viscosity: 713 cP (20 °C
- SUM Formula: C27H54F6N2O4S2
- Molecular Weight: 648.85
- Melting Point: -39 °C
- Density: 1.15 g/cm³
- ECW: 5.1 V
- Viscosity: 713 cP (20 °C
Methyltrioctylammonium bis(trifluoromethylsulfonyl)imide, CAS: 375395-33-8
Key Applications:
Extraction and Separation Processes
MTOA‑TFSI’s hydrophobicity, large organic cation, and weakly coordinating TFSI anion make it a high‑performance phase‑transfer medium for liquid–liquid extraction systems.
- Efficient extraction of metal ions, including lanthanides, actinides, and transition metals.
- Selective separation of hydrophobic organic molecules due to tunable solvation environments.
- Use as an ionic liquid diluent or co‑solvent in biphasic extraction systems to enhance distribution ratios and reduce solvent losses.
- Improved thermal and chemical stability compared to conventional quaternary ammonium extractants.
Metallic Fuel Reprocessing
MTOA‑TFSI supports advanced nuclear fuel cycle operations where ionic liquids are used to replace volatile organic solvents.
- Serves as a stable ionic medium for electrorefining and electrochemical separation of metallic nuclear fuels.
- Compatible with high‑temperature molten‑salt or ionic‑liquid-based processes for actinide partitioning.
- Reduces radiolytic degradation relative to chloride‑based systems, improving process longevity.
- Enables selective recovery of uranium, plutonium, and minor actinides in next‑generation pyroprocessing workflows.
Electrochemical Systems
The bulky, charge‑delocalized TFSI anion provides high ionic mobility and wide electrochemical stability.
- Electrolyte component for high‑voltage supercapacitors and batteries.
- Ionic liquid medium for electrodeposition of metals and alloys.
- Additive for improving interfacial stability in lithium and sodium metal systems.
Catalysis and Organic Synthesis
MTOA‑TFSI functions as a non‑volatile, thermally robust reaction medium.
- Solvent or co‑solvent for transition‑metal catalysis, including C–C and C–N bond‑forming reactions.
- Phase‑transfer catalyst for nucleophilic substitutions and alkylation reactions.
- Medium for biphasic catalytic systems requiring strong hydrophobicity and low coordinating ability.
Materials Processing
The long‑chain quaternary ammonium cation imparts plasticizing and structuring effects in polymer and composite systems.
- Ionic liquid additive for improving flexibility and conductivity in polymer electrolytes.
- Processing aid for nanoparticle dispersion and surface modification.
- Component in ionogels and hybrid materials requiring low volatility and high thermal stability.
High‑Temperature and Vacuum Applications
The TFSI anion’s exceptional thermal stability supports demanding operational environments.
- Heat‑transfer and thermal‑storage media in specialized industrial systems.
- Lubrication and anti‑wear additive for high‑vacuum or high‑temperature equipment.
- Non‑flammable alternative to conventional organic fluids in controlled‑atmosphere processes.
Please contact us if you want to learn more or need assistance with your order.