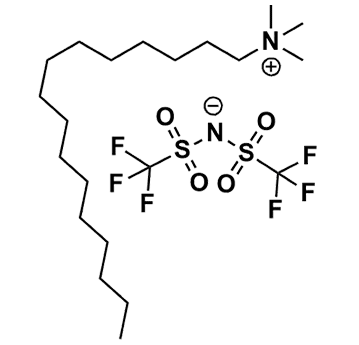
Hexadecyltrimethylammonium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $120.27 through $6,293.12
Product Code: IL-0327-HPCAS NO: 1031250-01-7
- Chemical Formula: C21H42F6N2O4S2
- Synonyms: N111(16) BTA, N111(16) NTf 2 , N111(16) TFSI, N111(16) BTI
- Weakly-coordinating anion
- Hydrophobic
SUM Formula: C21H42F6N2O4S2
Molecular Weight: 564.69
Melting Point: >RT
Purity: >99%
- SUM Formula: C21H42F6N2O4S2
- Molecular Weight: 564.69
- Melting Point: >RT
Hexadecyltrimethylammonium bis(trifluoromethylsulfonyl)imide, CAS: 1031250-01-7
Key Applications:
Surfactants, Self‑Assembly & Soft Materials
- Functions as a cationic surfactant with strong hydrophobic character, enabling micelle, vesicle, and bilayer formation in aqueous and mixed‑solvent systems.
- Supports nanostructured soft‑matter design, including templated mesoporous materials and controlled self‑assembly of amphiphilic systems.
- Used in colloidal stabilization and interfacial engineering where low water activity and high thermal stability are required.
Electrochemical Systems & Ionic Interfaces
- Serves as a hydrophobic ionic liquid surfactant for modifying electrode surfaces and tuning double‑layer behavior.
- Employed in electrodeposition, interfacial charge‑transfer studies, and ion‑transport modulation in low‑polarity media.
- Useful in non‑aqueous electrolyte formulations where long‑chain cations enhance viscosity, interfacial adhesion, or film formation.
Phase‑Transfer & Catalysis
- Acts as a phase‑transfer catalyst for reactions involving hydrophobic substrates or biphasic organic/ionic liquid systems.
- Enhances reaction rates and selectivity in nucleophilic substitutions, oxidations, and metal‑catalyzed transformations requiring strong ion pairing.
Antimicrobial & Biochemical Systems
- Long‑chain quaternary ammonium cations exhibit broad antimicrobial activity, enabling use in disinfectant formulations, membrane‑disruption studies, and controlled‑release systems.
- Supports biophysical investigations of lipid–surfactant interactions and protein denaturation in low‑water environments.
Materials Science & Nanostructure Control
- Used as a structure‑directing agent in nanoparticle synthesis, including metal, metal oxide, and semiconductor nanomaterials.
- Facilitates surface functionalization of nanoparticles and polymers, improving dispersion in hydrophobic matrices.
- Supports fabrication of ionically conductive polymer blends and hybrid organic–inorganic materials.
Thermal & Chemical Stability Applications
- The TFSI⁻ anion provides exceptional thermal and electrochemical stability, enabling use in high‑temperature surfactant systems, molten‑salt analogues, and solvent‑free ionic assemblies.
- Suitable for low‑volatility formulations and high‑performance lubricants where ionic amphiphiles improve boundary‑layer behavior.
Please contact us if you want to learn more or need assistance with your order.