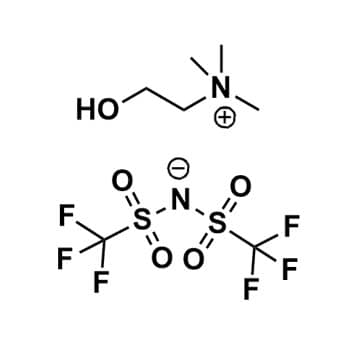
Choline bis(trifluoromethylsulfonyl)imide, >99%
Price range: $155.75 through $8,208.21
Product Code: IL-0110-HPCAS NO: 827027-25-8
- Chemical Fr: C7H14F6N2O4S2
- Synonyms: Chol BTA, Chol NTf2, Chol TFSI, Chol BTI, N,N,N-thimethyl-N-(2-hydroxyethyl) ammonium bis(trifluoromethylsulfonyl)imide, 1,1,1-thimethyl-1-(2-hydroxyethyl) ammonium bis(trifluoromethylsulfonyl)imide
- Weakly-coordinating anion
- Hydrophobic
Conductivity: 3.98 mS/cm (45 °C)
SUM Formula: C7H14F6N2O4S2
Molecular Weight: 368.32
Melting Point: 35-37 °C
Density: 1.508 (25 °C)
Purity: >99%
Viscosity: 49.5 cP (45 °C)
- SUM Formula: C7H14F6N2O4S2
- Molecular Weight: 368.32
- Melting Point: 35-37 °C
- Density: 1.508 (25 °C)
- Viscosity: 49.5 cP (45 °C)
Choline bis(trifluoromethylsulfonyl)imide, CAS: 827027-25-8
Key Applications:
Electrolytes for Energy Storage
- Serves as a high‑stability ionic liquid electrolyte component in lithium‑ion, sodium‑ion, and emerging multivalent battery systems.
- Enhances ionic conductivity and electrochemical window in solid–liquid hybrid electrolytes.
- Used in polymer gel electrolytes and ionogel matrices for flexible and solid‑state devices.
Electrochemical Devices & Interfaces
- Employed in supercapacitor electrolytes to improve thermal stability and long‑term cycling performance.
- Supports electrode surface engineering, including controlled SEI formation and interfacial ion transport studies.
- Utilized in redox‑active systems for flow batteries and electrocatalytic platforms.
Catalysis & Green Chemistry
- Functions as a thermally robust, non‑volatile reaction medium for acid‑ and base‑catalyzed transformations.
- Applied in transition‑metal catalysis, including cross‑coupling, hydrogenation, and CO₂ conversion reactions.
- Enables biphasic catalysis and catalyst recycling due to its immiscibility with many organic solvents.
Separation Science & Extraction
- Used in liquid–liquid extraction of metal ions, particularly rare‑earth and transition‑metal species, due to strong TFSI⁻ coordination.
- Supports selective extraction of hydrophobic organics and fluorinated compounds.
- Applied in membrane‑based separations and supported ionic liquid phases (SILPs).
Materials Science & Polymer Engineering
- Incorporated into polymer matrices to tune conductivity, mechanical flexibility, and thermal behavior.
- Used in ionogels, block copolymers, and advanced dielectric materials.
- Serves as a plasticizer or ionic dopant in conductive polymers and elastomers.
Surface & Colloid Science
- Acts as a surfactant‑like ionic liquid for stabilizing dispersions of nanoparticles, carbon materials, and metal oxides.
- Supports controlled self‑assembly and nanostructure formation in soft materials.
Analytical & Spectroscopic Applications
- Provides a low‑volatility, high‑purity medium for NMR, FTIR, and electrochemical characterization.
- Used in fundamental studies of ion pairing, solvation, and transport phenomena.
Contact us if you want to learn more or need assistance with your order.