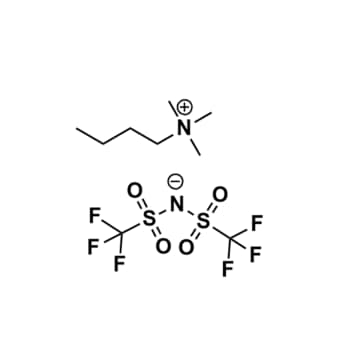
Butyltrimethylammonium bis(trifluoromethylsulfonyl)imide, >99.5%
Price range: $192.46 through $1,602.36
Product Code: IL-0032-UPCAS NO: 258273-75-5
- Chemical Formula: C9H18F6N2O4S2
- Synonyms: N1114 BTA, N1114 NTf 2 , N1114 TFSI, N1114 BTI
- Weakly-coordinating anion
- Hydrophobic
For High-Purity of >99% Purity, refer to IL-0032-HP
Conductivity: 2.86 mS/cm
SUM Formula: C9H18F6N2O4S2
Molecular Weight: 396.37
Melting Point: 7 °C
Density: 1.40 g/cm³
ECW: 6.1 V
Purity: >99.%%
Viscosity: 99.5 cP
- SUM Formula: C9H18F6N2O4S2
- Molecular Weight: 396.37
- Melting Point: 7 °C
- Density: 1.40 g/cm³
- ECW: 6.1 V
- Viscosity: 99.5 cP
Butyltrimethylammonium bis(trifluoromethylsulfonyl)imide, CAS: 258273-75-5
Key Applications:
Electrochemical Systems
- Serves as a high‑stability supporting electrolyte in non‑aqueous electrochemical cells, including lithium, sodium, and multivalent metal systems.
- Enhances ionic conductivity and suppresses parasitic reactions in high‑voltage environments due to the TFSI anion’s oxidative robustness.
- Used in electrodeposition studies where controlled ion mobility and low water content are essential.
Battery and Energy Storage Research
- Functions as an additive or co‑salt in electrolyte formulations for next‑generation batteries, improving SEI formation and cycling stability.
- Supports research on ionic‑liquid‑based electrolytes for solid‑state and hybrid solid–liquid systems.
- Applied in redox‑flow battery development where wide electrochemical windows and thermal stability are required.
Materials Science and Polymer Processing
- Acts as a dopant or ionic modifier in polymer matrices, enabling tunable conductivity in ion gels, block copolymers, and elastomeric materials.
- Used in the preparation of polymer electrolytes and membranes with enhanced mechanical and electrochemical performance.
- Facilitates ion‑exchange processes in advanced materials where hydrophobic, weakly coordinating anions are advantageous.
Catalysis and Synthesis
- Employed as a phase‑transfer or ionic‑medium component in organic synthesis, particularly under low‑volatility or solvent‑free conditions.
- Supports catalytic systems that benefit from the TFSI anion’s non‑coordinating nature, improving catalyst stability and turnover.
- Useful in halide‑free reaction environments where minimal nucleophilicity is required.
Spectroscopy and Fundamental Studies
- Provides a well‑defined cation–anion pair for benchmarking ion transport, solvation, and interfacial behavior in ionic liquid research.
- Used in vibrational, NMR, and dielectric spectroscopy to study ion pairing and dynamic relaxation processes.
- Serves as a model salt for computational and theoretical investigations of ionic liquids with quaternary ammonium cations.
Please contact us if you want to learn more or need assistance with your order.