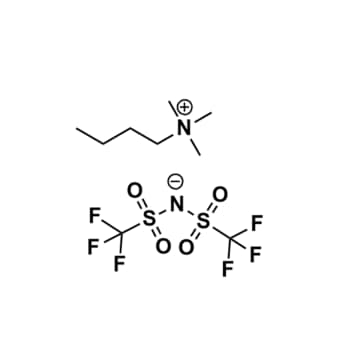
Butyltrimethylammonium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $134.49 through $7,432.55
Product Code: IL-0032-HPCAS NO: 258273-75-5
- Chemical Formula: C9H18F6N2O4S2
- Synonyms: N1114 BTA, N1114 NTf2, N1114 TFSI, N1114 BTI
- Weakly-coordinating anion.
- Hydrophobic
For Ultra-High Purity of >99.5% refer to IL-0032-UP
Conductivity: 2.86 mS/cm
SUM Formula: C9H18F6N2O4S2
Molecular Weight: 396.37 g/mol
Melting Point: 7 °C
Density: 1.40 g/cm³
ECW: 6.1 V
Purity: >99%
Viscosity: 99.5 cP @25 °C
- SUM Formula: C9H18F6N2O4S2
- Molecular Weight: 396.37 g/mol
- Melting Point: 7 °C
- Density: 1.40 g/cm³
- ECW: 6.1 V
- Viscosity: 99.5 cP @25 °C
Butyltrimethylammonium bis(trifluoromethylsulfonyl)imide, CAS: 258273-75-5
Key Applications:
1. Electrochemical & Energy Storage Applications
- Supporting electrolyte in non‑aqueous systems
Enhances ionic conductivity and widens the electrochemical stability window in organic solvents.
- Lithium‑ion and sodium‑ion battery research
Used as an additive or co‑salt to improve SEI formation, suppress dendrites, and stabilize high‑voltage cathodes.
- Supercapacitors and hybrid capacitors
Provides high ionic mobility and low viscosity when paired with compatible solvents or ionic liquid matrices.
- Electrodeposition media
Enables smooth, uniform metal deposition due to its low coordinating TFSI anion.
2. Catalysis & Synthetic Chemistry
- Phase‑transfer catalysis
The quaternary ammonium cation facilitates transport of reactive species across immiscible phases.
- Non‑coordinating reaction medium
TFSI’s weak coordination supports reactions requiring highly reactive cations or electrophiles.
- Organometallic stabilization
Useful for generating stable ionic complexes in low‑nucleophilicity environments.
3. Materials Science & Soft Matter
- Polymer electrolytes
Incorporated into polymer matrices (PEO, PMMA, PVDF) to enhance ionic transport and mechanical flexibility.
- Ion gels and solid-state ionic materials
Forms stable, low‑volatility ionogels for flexible electronics and sensors.
- Antistatic and charge‑control additives
The quaternary ammonium headgroup provides persistent surface charge control in coatings and films.
4. Thermal & Transport Applications
- Heat‑transfer and thermal‑stability studies
High thermal stability of TFSI salts makes them useful in benchmarking ionic liquid performance.
- Solvent‑free ionic media
BTMA‑TFSI can act as a component in eutectic ionic mixtures to tune viscosity, conductivity, and melting point.
5. Separation Science
- Ion‑pairing reagent in chromatography
Enhances separation of polar analytes in HPLC and ion chromatography.
- Extraction and purification systems
Hydrophobic TFSI anion supports selective extraction of metal ions and organic species.
6. Environmental & Industrial Chemistry
- Electrochemical CO₂ reduction studies
Provides a stable, low‑nucleophilicity medium for probing catalytic pathways.
- Corrosion‑resistant ionic environments
TFSI salts are used in testing metal stability under extreme conditions.
Please contact us if you want to learn more or need assistance with your order.