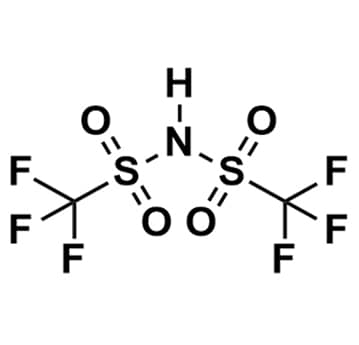
Bis(trifluoromethylsulfonyl)imide, 70% aqueous solution, >99%
Price range: $89.12 through $2,713.41
Product Code: KI-0055-HPCAS NO: 82113-65-3
- Chemical Formula: C2HF6NO4S2
- Synonyms: HBTA, HTFSI, HNTf 2
To order 5kg or more, please email us at info@roco.global
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C2HF6NO4S2
Molecular Weight: 281.16
Purity: >99%
- SUM Formula: C2HF6NO4S2
- Molecular Weight: 281.16
Bis(trifluoromethylsulfonyl)imide, CAS: 82113-65-3
Key Applications:
-
- Reduced fuming and safer handling compared to anhydrous HTFSI.
- Improved metering accuracy for neutralization reactions.
- Lower volatility and easier integration into continuous‑flow or batch salt‑production systems.
- Compatible with industrial‑scale purification and crystallization workflows for downstream TFSI⁻ salts.
Electrolyte Manufacturing and Salt Precursor Processing
- Used as a controlled‑strength precursor for generating TFSI⁻ salts through neutralization with lithium, sodium, potassium, ammonium, phosphonium, or imidazolium bases.
- Aqueous format enables safer handling and more uniform stoichiometric control during salt formation, minimizing thermal load and avoiding direct handling of anhydrous HTFSI.
- Supports scalable production of battery‑grade salts, ionic liquids, and deep eutectic electrolytes.
Polymer and Membrane Functionalization
- Acts as a sulfonylating and ion‑exchange reagent for preparing TFSI‑functionalized polymers, ionomers, and proton‑conducting membranes.
- Aqueous medium improves compatibility with hydrophilic polymer backbones and reduces the need for aggressive organic solvents.
Catalysis and Acid‑Mediated Transformations
- Serves as a strong Brønsted acid for aqueous or biphasic catalytic systems requiring high acidity without introducing halides.
- Enables acid‑catalyzed esterifications, rearrangements, and dehydration reactions where non‑nucleophilic counterions are essential.
Surface Treatment and Materials Processing
- Used to introduce TFSI⁻ into inorganic and hybrid materials through ion‑exchange or protonation pathways.
- Supports preparation of TFSI‑doped metal oxides, conductive coatings, and surface‑modified nanoparticles.
Electrochemical and Analytical Applications
- Provides a controlled route to generate TFSI‑based electrolytes for supercapacitors, redox‑flow systems, and specialty electrochemical cells.
- Aqueous form simplifies pH adjustment and ion‑exchange workflows in analytical chemistry and materials screening.
Advantages of the Aqueous 70–72% Format
- Reduced fuming and safer handling compared to anhydrous HTFSI.
- Improved metering accuracy for neutralization reactions.
- Lower volatility and easier integration into continuous‑flow or batch salt‑production systems.
- Compatible with industrial‑scale purification and crystallization workflows for downstream TFSI⁻ salts.
Please contact us if you want to learn more or need assistance with your order.