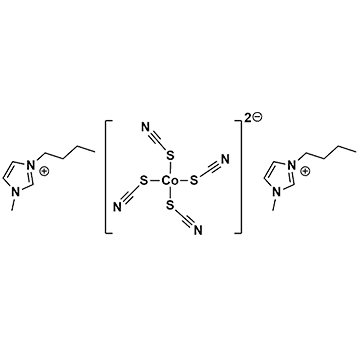
Bis(1-butyl-3-methylimidazolium) tetrathiocyanatocobaltate, >99%
Price range: $171.19 through $2,743.16
Product Code: IL-0310-HPCAS NO: 1245942-47-5
- Chemical Formula: C20H30CoN8S4
- Synonyms: BMIM2 Co(SCN)4 , C1 C4 Im2 Co(SCN)4
- Aromatic
SUM Formula: C20H30CoN8S4
Molecular Weight: 569.70
Melting Point: <RT
Density: 1.218 g/cm³ (26 °C)
Purity: >99%
Viscosity: 268.6 cP (25 °C)
- SUM Formula: C20H30CoN8S4
- Molecular Weight: 569.70
- Melting Point:
- Density: 1.218 g/cm³ (26 °C)
- Viscosity: 268.6 cP (25 °C)
Bis(1-butyl-3-methylimidazolium) tetrathiocyanatocobaltate, CAS: 1245942-47-5
Key Applications:
Advanced Electrochemical Systems
- Serves as a redox‑active ionic liquid with a cobalt‑based tetrathiocyanate anion, enabling tunable electron‑transfer behavior in electrochemical devices.
- Used in the development of non‑volatile electrolytes for high‑temperature or high‑stability energy‑storage platforms.
- Supports research into ionic‑liquid‑based redox mediators for dye‑sensitized solar cells and next‑generation electrochemical sensors.
Coordination Chemistry and Catalysis
- Provides a stable, well‑defined cobalt coordination environment useful for studying ligand‑field effects, electron‑donor/acceptor behavior, and metal–ligand charge transfer.
- Employed as a functional ionic medium for catalytic transformations where transition‑metal centers and soft‑donor ligands enhance reaction selectivity.
- Enables mechanistic studies of thiocyanate‑rich cobalt complexes in homogeneous catalysis.
Materials Science and Functional Thin Films
- Utilized in the fabrication of conductive or magnetically responsive thin films due to the paramagnetic cobalt center and ionic‑liquid matrix.
- Supports research into hybrid organic–inorganic materials where metal–thiocyanate clusters contribute to tunable optical or electronic properties.
- Investigated for incorporation into polymer–ionic liquid composites to enhance ionic conductivity and mechanical stability.
Spectroscopy, Magnetism, and Fundamental Studies
- Serves as a model compound for probing cobalt(II) electronic structure using UV–vis, EPR, and vibrational spectroscopy.
- Used in magnetic‑susceptibility studies to understand spin‑state behavior in thiocyanate‑coordinated cobalt complexes.
- Provides a stable, low‑volatility medium for studying ion pairing, solvation, and structural organization in imidazolium‑based ionic liquids.
Solvent and Extraction Research
- Functions as a task‑specific ionic liquid for selective extraction of soft‑donor metal ions due to the tetrathiocyanate ligand environment.
- Explored in biphasic separation systems where metal–ligand interactions drive selective partitioning.
- Supports studies on metal‑ion transport, coordination equilibria, and solvent structuring in ionic‑liquid phases.
Please contact us if you want to learn more or need assistance with your order.