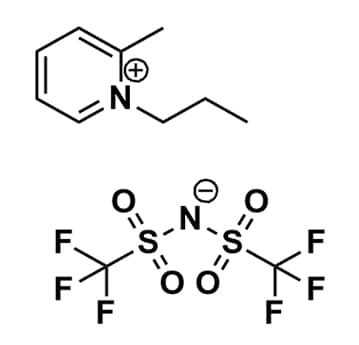
2-Methyl-1-propylpyridinium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $179.73 through $9,858.65
Product Code: IL-0248-HPCAS NO: 1456877-99-8
- Chemical Formula: C11H14F6N2O4S2
- Synonyms: 1-Propyl-2-picolinium bis(trifluoromethylsulfonyl)imide, N-Propyl-2-methylpyridinium bis(trifluoromethylsulfonyl)imide, Pro2Pic BTA, Pro-2-Pic BTA, Pro2Pic NTf2, Pro2Pic TFSI, Pro2Pic BTI
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 1.31 mS/cm (25 °C)
SUM Formula: C11H14F6N2O4S2
Molecular Weight: 416.36
Melting Point: <RT
Purity: >99%
Viscosity: 81.6 cP (25 °C)
- SUM Formula: C11H14F6N2O4S2
- Molecular Weight: 416.36
- Melting Point:
- Viscosity: 81.6 cP (25 °C)
2-Methyl-1-propylpyridinium bis(trifluoromethylsulfonyl)imide, CAS: 1456877-99-8
Key Applications:
Electrochemical Systems
- High‑stability electrolyte component for lithium, sodium, and multivalent metal batteries, leveraging the TFSI⁻ anion’s wide electrochemical window.
- Ionic‑liquid medium for supercapacitors requiring low volatility and strong thermal resilience.
- Conductive additive in redox‑flow batteries to enhance ion mobility and suppress side reactions.
Thermal & Chemical Stability Applications
- Heat‑transfer and thermal‑management fluids in high‑temperature or vacuum environments, where non‑flammability and low vapor pressure are essential.
- Replacement for volatile organic solvents in high‑temperature synthesis, catalytic cycles, and controlled‑atmosphere reactions.
Catalysis & Organic Synthesis
- Reaction medium for transition‑metal catalysis, including C–C and C–N bond‑forming reactions that benefit from ionic‑liquid stabilization.
- Solvent for selective extraction, biphasic catalysis, and phase‑transfer processes, especially where hydrophobicity and chemical inertness are required.
Separation Science & Extraction
- Hydrophobic ionic liquid for liquid–liquid extraction of metal ions, hydrophobic organics, and fluorinated species.
- Tunable medium for chromatographic method development, particularly in ion‑pairing or non‑aqueous separations.
Materials Science & Polymer Engineering
- Additive for ion‑conducting polymers, gels, and elastomers to improve conductivity, flexibility, and thermal stability.
- Processing aid in membrane fabrication, enabling controlled microstructure and enhanced transport properties.
Surface Science & Nanotechnology
- Stabilizing medium for nanoparticle synthesis, preventing aggregation and enabling narrow size distributions.
- Ionic‑liquid phase for surface modification, thin‑film deposition, and interfacial engineering.
Tribology & Lubrication
- High‑performance lubricant or lubricant additive for extreme‑pressure and high‑temperature environments, benefiting from TFSI⁻‑based ionic liquids’ low wear and friction coefficients.
Please contact us if you want to learn more or need assistance with your order.