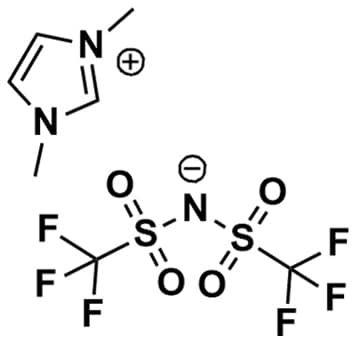
1,3-Dimethylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $142.89 through $8,106.36
Product Code: IL-0198-HPCAS NO: 174899-81-1
- Chemical Formula: C7H9F6N3O4S2
- Synonyms: DiMIM BTA, DiMIM NTf 2 , DiMIM TFSI, DiMIM BTI, Im 11 BTA
- Weakly-coordinating anion
- Aromatic
Conductivity: 9.36 mS/cm (30 °C)
SUM Formula: C7H9F6N3O4S2
Molecular Weight: 377.28
Melting Point: 22 °C
Density: 1.57 g/cm³ (22 °C)
ECW: 3.2V
Purity: >99%
Viscosity: 37.1 cP (25°C)
- SUM Formula: C7H9F6N3O4S2
- Molecular Weight: 377.28
- Melting Point: 22 °C
- Density: 1.57 g/cm³ (22 °C)
- ECW: 3.2V
- Viscosity: 37.1 cP (25°C)
1,3-Dimethylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 174899-81-1
Key Applications:
Electrochemical Systems
- High‑performance electrolyte component for lithium‑ion, sodium‑ion, and magnesium‑ion batteries, where its low viscosity and high ionic conductivity support efficient ion transport.
- Used in supercapacitors and electrochemical double‑layer capacitors (EDLCs) to enhance cycling stability and widen the electrochemical window.
- Suitable for electrodeposition of reactive or air‑sensitive metals due to its thermal stability and non‑volatile nature.
Catalysis & Organic Synthesis
- Employed as a non‑coordinating reaction medium for transition‑metal catalysis, enabling improved selectivity and turnover.
- Supports acid‑catalyzed and base‑catalyzed transformations, including alkylation, esterification, and cyclization reactions.
- Utilized in biocatalysis as a co‑solvent that stabilizes enzymes and enhances substrate solubility.
Separation Science & Extraction
- Effective in liquid–liquid extraction of metal ions, especially rare‑earth and actinide species, due to the hydrophobic TFSI⁻ anion.
- Applied in gas absorption and separation, including CO₂ capture, where its tunable solvation properties improve uptake efficiency.
- Used in chromatographic stationary phases to enhance selectivity for polar and aromatic analytes.
Materials Science & Polymer Engineering
- Incorporated into polymer electrolytes and ion gels to improve mechanical flexibility and ionic transport.
- Acts as a plasticizer in high‑performance polymers, enhancing conductivity and thermal stability.
- Supports fabrication of ionic‑liquid‑based membranes for fuel cells, gas separation, and pervaporation.
Thermal & Process Engineering
- Serves as a heat‑transfer and thermal‑storage medium in high‑temperature environments due to its exceptional thermal stability.
- Used as a lubricant additive to reduce friction and wear in demanding mechanical systems.
- Functions as a stable solvent for high‑temperature or vacuum‑sensitive processes where conventional organics fail.
Please contact us if you want to learn more or need assistance with your order.