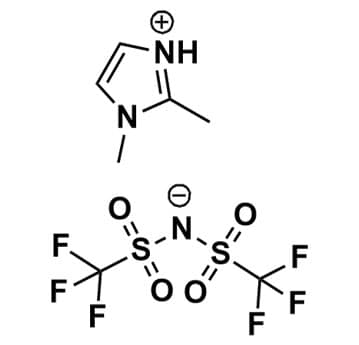
1,2-Dimethylimidazolium bis(trifluoromethylsulfonyl)imide, >98%
Price range: $260.45 through $2,063.17
Product Code: IL-0278-SGCAS NO: 353239-12-0
- Chemical Fr: C7H9F6N3O4S2
- Synonyms: DiMIM BTA, DiMIM NTf2, DiMIM TFSI, DiMIM BTI
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
- Protic
To order 5kg or more, please email us at info@roco.global
**This product will incur a $97.00 HazMat fee when the order is placed.**
Conductivity: 1.08 mS/cm (25 °C)
SUM Formula: C7H9F6N3O4S2
Molecular Weight: 377.28
Melting Point: <RT
Purity: >98%
Viscosity: 106 cP (25 °C)
- SPECIFIC GRAVITY: NA
- SUM Formula: C7H9F6N3O4S2
- Molecular Weight: 377.28
- Melting Point:
- Viscosity: 106 cP (25 °C)
1,2-Dimethylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 353239-12-0
1. Electrolytes for High‑Performance Energy Storage
- Functions as a low‑viscosity ionic liquid electrolyte component for Li‑ion, Na‑ion, and Mg‑ion batteries.
- The TFSI⁻ anion provides wide electrochemical stability windows suitable for high‑voltage cathodes.
- Supports ionic‑liquid‑based gel polymer electrolytes requiring enhanced ion mobility.
2. Supercapacitors and Hybrid Capacitors
- Serves as a stable ionic medium for EDLCs and pseudocapacitors with high ionic conductivity and thermal stability.
- The 1,2‑dimethyl substitution reduces crystallinity, improving low‑temperature performance.
3. Solvent and Co‑solvent for Organic Synthesis
- Acts as a non‑volatile, thermally robust reaction medium for transition‑metal catalysis, C–C coupling, and heterocycle functionalization.
- The cation’s steric and electronic profile can improve selectivity in organometallic transformations.
4. Extraction and Separation Processes
- Effective in liquid–liquid extraction of metal ions due to the hydrophobic TFSI⁻ anion.
- Useful in CO₂ capture systems where imidazolium ionic liquids enhance absorption capacity and tunability.
5. Electrochemical Deposition and Surface Engineering
- Provides a stable ionic environment for metal electrodeposition, including aluminum, rare‑earth metals, and reactive alloys.
- Supports smooth, dendrite‑resistant deposition due to low water content and high thermal stability.
6. Polymer Processing and Advanced Materials
- Functions as a plasticizer or ionic dopant in conductive polymers such as PEDOT and polyaniline.
- Enhances mechanical flexibility and ionic conductivity in ionogels and polymer electrolytes.
7. Spectroscopy and Fundamental Research
- Used as a model ionic liquid for studying cation–anion interactions, solvation dynamics, and charge transport.
- The 1,2‑dimethyl substitution pattern provides a distinct benchmark for structure–property correlation studies.
Contact us if you want to learn more or need assistance with your order.