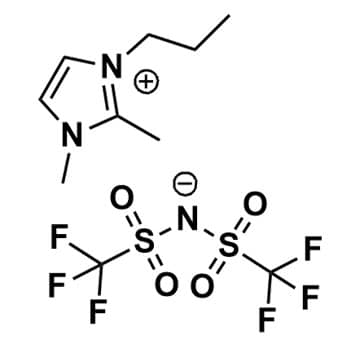
1,2-Dimethyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $179.73 through $9,858.65
Product Code: IL-0134-HPCAS NO: 169051-76-7
- Chemical Formula: C10H15F6N3O4S2
- Synonyms: DMPIIm, PDiMIM BTA, PDiMIM NTf2, PDiMIM TFSI, PDiMIM BTI, IM113 BTA
- Weakly-coordinating anion
- Aromatic
- Hydrophobic
Conductivity: 1.95 mS/cm (25 °C)
SUM Formula: C10H15F6N3O4S2
Molecular Weight: 419.36
Melting Point: 15 °C
Density: 1.45 g/cm³ (25 °C)
ECW: 4.6 V
HMIS Key: NA
Purity: >99%
Viscosity: 107 cP (25 °C)
- SPECIFIC GRAVITY: NA
- SUM Formula: C10H15F6N3O4S2
- Molecular Weight: 419.36
- Melting Point: 15 °C
- Density: 1.45 g/cm³ (25 °C)
- ECW: 4.6 V
- HMIS KEY: NA
- TSCA: NA
- Viscosity: 107 cP (25 °C)
1,2-Dimethyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 169051-76-7
Key Applications
Electrochemical Systems
- Serves as a high‑stability ionic liquid electrolyte component for lithium, sodium, and multivalent metal batteries, benefiting from the TFSI⁻ anion’s wide electrochemical window and low coordinating character.
- Supports high‑temperature supercapacitor formulations where low volatility and strong thermal resilience are required.
- Functions as a conductive medium in redox‑flow and hybrid flow battery research, particularly where cation structure influences viscosity and ion mobility.
Catalysis and Synthesis
- Acts as a non‑volatile reaction medium for transition‑metal‑catalyzed transformations, including C–C and C–N coupling, hydrogenation, and selective oxidations.
- Provides a tunable solvent environment for organocatalysis, enabling enhanced selectivity and recyclability.
- Useful in biphasic catalysis where phase separation and catalyst retention are critical.
Materials Science and Polymer Processing
- Employed as a plasticizer or ionic additive in polymer electrolytes, ionogels, and high‑performance elastomers.
- Facilitates dispersion and stabilization of nanoparticles, carbon materials, and metal oxides due to its balanced polarity and low nucleophilicity.
- Supports surface modification and thin‑film deposition processes where ionic liquids enable controlled morphology and reduced defect formation.
Separation Science and Extraction
- Functions as an extraction medium for metal ions, hydrophobic organics, and fluorinated species, leveraging the hydrophobic TFSI⁻ anion.
- Enables selective partitioning in liquid–liquid extraction systems, particularly for rare‑earth and transition‑metal separations.
- Useful in gas absorption studies, including CO₂ capture, where cation substitution patterns influence solubility and transport.
Thermal and Chemical Stability Applications
- Suitable for high‑temperature lubrication and tribological studies where non‑flammability and thermal endurance are required.
- Serves as a stable medium for studying reaction kinetics under elevated temperatures or inert‑atmosphere conditions.
- Supports high‑vacuum and high‑temperature environments where conventional solvents degrade or volatilize.
Analytical and Spectroscopic Uses
- Provides a low‑volatility matrix for electrochemical impedance spectroscopy, NMR diffusion studies, and vibrational spectroscopy.
- Useful in calibrating ion transport models due to its well‑defined cation structure and predictable physicochemical behavior.
Please contact us if you want to learn more or need assistance with your order.