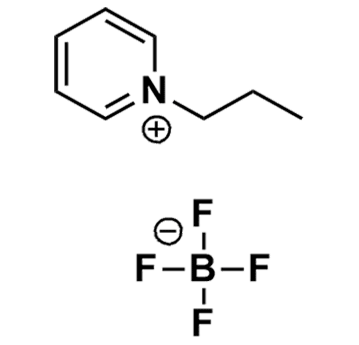
1-Propylpyridinium tetrafluoroborate, >98%
Price range: $260.45 through $2,549.21
Product Code: IL-0252-HPCAS NO: 239084-00-5
- Chemical Formula: C8H12BF4N
- Synonyms: ProPy BF 4 , N-Propylpyridinium tetrafluoroborate
- Anion decomposes slowly in the presence of water.
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 1.35 mS/cm
SUM Formula: C8 H12 BF4 N
Molecular Weight: 208.99
Melting Point: 12 °C
Density: 1.246 g/cm3
Purity: >98%
Viscosity: 137.9 cP
- SUM Formula: C8 H12 BF4 N
- Molecular Weight: 208.99
- Melting Point: 12 °C
- Density: 1.246 g/cm3
- Viscosity: 137.9 cP
1-Propylpyridinium tetrafluoroborate, CAS: 239084-00-5
Key Applications:
Electrochemical Systems
- Functions as a stable, low‑volatility ionic medium for electrochemical studies requiring well‑defined ion transport behavior.
- Supports electrodeposition experiments where moderate viscosity and wide electrochemical windows are advantageous.
- Useful as an electrolyte component in prototype capacitors and redox‑active device testing.
Catalysis and Organic Synthesis
- Serves as a non‑volatile reaction medium for acid‑sensitive or moisture‑sensitive transformations.
- Enhances selectivity in biphasic and phase‑transfer‑assisted reactions due to its tunable polarity and ionic character.
- Provides a controlled environment for metal‑catalyzed processes, including cross‑coupling and hydrogenation studies.
Materials Science and Thin‑Film Processing
- Employed as a solvent or co‑solvent in the preparation of polymer electrolytes, ion‑conductive films, and hybrid materials.
- Supports nanoparticle dispersion and stabilization, particularly for metal and metal‑oxide systems.
- Useful in templating and morphology‑control studies for porous materials and ionic polymer composites.
Separation Science
- Acts as a mobile‑phase additive or stationary‑phase modifier in liquid chromatography to improve peak shape and ion‑pairing behavior.
- Enhances selectivity in extraction systems involving metal ions or polar organics.
Thermal and Physical Property Research
- Suitable for fundamental studies of ionic liquid structure–property relationships, including viscosity, conductivity, and ion‑pairing dynamics.
- Used in benchmarking experiments comparing pyridinium‑based ionic liquids with varying alkyl chain lengths.
Please contact us if you want to learn more or need assistance with your order.