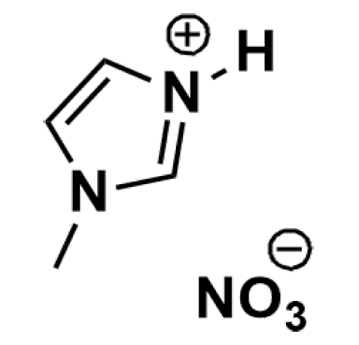
1-Methylimidazolium nitrate, >98%
Price range: $376.52 through $803.57
Product Code: IL-0264-SGCAS NO: 156204-43-2
- Chemical Formula: C4H7N3O3
- Synonyms: MIM NO3, Methylimidazolium nitrate, 3-methylimidazolium nitrate
- Aromatic
- Hydrophilic
- Protic
To order 5kg or more, please email us at info@roco.global
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C4H7N3O3
Molecular Weight: 145.12
Melting Point: 66°C
Purity: >98%
- SUM Formula: C4H7N3O3
- Molecular Weight: 145.12
- Melting Point: 66°C
1-Methylimidazolium nitrate, CAS: 156204-43-2
Key Applications:
1. Solvent and Reaction Medium
- Functions as a high‑polarity solvent for acid‑catalyzed and hydrogen‑bond‑driven transformations, including esterifications, nitrations, and dehydration reactions.
- Supports protic reaction environments, enabling enhanced rates in transformations that benefit from proton transfer or hydrogen‑bond stabilization.
- Useful for transition‑metal catalysis, where its coordinating yet compact structure minimizes steric hindrance around catalytic centers.
2. Hydrogen‑Bonding and Supramolecular Chemistry
- Exhibits strong hydrogen‑bond donor and acceptor interactions, making it suitable for studying solvation dynamics, ion pairing, and molecular recognition.
- Applied in self‑assembly and supramolecular host–guest systems, where its protic nature modulates assembly pathways and stabilizes charged intermediates.
- Enables spectroscopic investigations of hydrogen‑bond networks due to its well‑defined vibrational signatures and tunable acidity.
3. Biomass and Carbohydrate Processing
- Effective for dissolving and swelling lignocellulosic materials, particularly when hydrogen bonding assists in disrupting cellulose crystallinity.
- Supports pretreatment and fractionation workflows for biomass upgrading, including hemicellulose extraction and lignin modification.
- Serves as a medium for carbohydrate derivatization, where its protic character enhances solubility and reactivity.
4. Electrochemical and Materials Research
- Acts as a protic ionic liquid electrolyte for low‑temperature electrochemical studies, benefiting from its ionic conductivity and hydrogen‑bond‑mediated proton mobility.
- Used in thin‑film deposition, polymer doping, and ion‑transport studies, where nitrate contributes to redox‑active behavior.
- Supports nanomaterial dispersion and surface modification, especially for metal oxides and carbonaceous materials.
5. Separation Science
- Functions as a selective extraction medium for metal ions, leveraging hydrogen bonding and nitrate coordination to tune selectivity.
- Applied in liquid–liquid extraction, supported liquid membranes, and ion‑exchange systems requiring high polarity and controlled acidity.
- Useful for gas absorption studies, including NOx and SOx capture, where nitrate interactions influence uptake.
6. Energetic Materials and Nitration Chemistry
- Serves as a safe, nonvolatile ionic medium for studying nitrate‑based energetic formulations.
- Supports nitration reactions where its protic, hydrogen‑bonding environment stabilizes reactive intermediates.
- Used in thermal decomposition and combustion studies due to its well‑characterized nitrate chemistry.
Please contact us if you want to learn more or need assistance with your order.