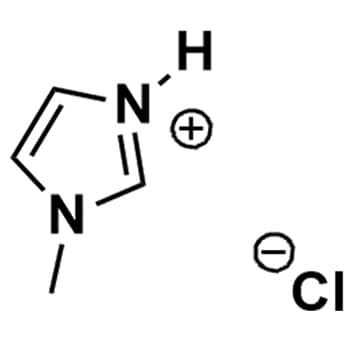
1-Methylimidazolium chloride, >98%
Price range: $89.12 through $503.49
Product Code: IL-0094-SGCAS NO: 35487-17-3
- Chemical Formula: C4H7ClN2
- Synonyms: MIM Cl
- Hydrophilic
- Aromatic
- Protic
To order 5kg or more, please email us at info@roco.global
** This product will incur a $97.00 HazMat fee when the order is placed. **
SUM Formula: C4H7ClN2
Molecular Weight: 118.57
Melting Point: >RT
Purity: >98%
- SUM Formula: C4H7ClN2
- Molecular Weight: 118.57
- Melting Point: >RT
1-Methylimidazolium chloride, CAS: 35487-17-3
Key Applications
1. Catalysis and Organic Synthesis
- Functions as a polar, non‑volatile medium for acid‑ and base‑catalyzed reactions.
- Supports nucleophilic substitution, alkylation, and condensation reactions where imidazolium cations stabilize transition states.
- Useful as a precursor for synthesizing N‑alkylated imidazolium ionic liquids via anion metathesis.
2. Electrochemical Systems
- Serves as a component in ionic‑liquid‑based electrolytes for low‑temperature electrochemical studies.
- Enhances ionic conductivity in mixed‑solvent systems due to the small, highly mobile cation.
- Applied in electrodeposition research where chloride coordination influences metal nucleation behavior.
3. Materials Science and Polymer Processing
- Acts as a solvent or co‑solvent for cellulose, chitin, and other hydrogen‑bond‑rich biopolymers.
- Enables homogeneous dissolution for membrane casting, fiber spinning, and derivatization studies.
- Used in templating and directing the morphology of inorganic and hybrid materials.
4. Phase Behavior and Solvation Studies
- Serves as a model imidazolium salt for studying ion pairing, hydrogen bonding, and solvation dynamics.
- Frequently used in computational and spectroscopic benchmarking due to its simple cation structure.
5. Precursor for Functional Ionic Liquids
- Provides a starting point for synthesizing task‑specific ionic liquids through anion exchange (e.g., BF₄⁻, PF₆⁻, NTf₂⁻).
- Supports development of ionic liquids tailored for CO₂ capture, catalysis, and electrochemical applications.
6. Biochemical and Extraction Applications
- Applied in biphasic extraction systems for selective separation of metal ions and organic compounds.
- Investigated as a medium for enzyme stabilization and activity modulation in non‑aqueous environments.
Please contact us if you want to learn more or need assistance with your order.