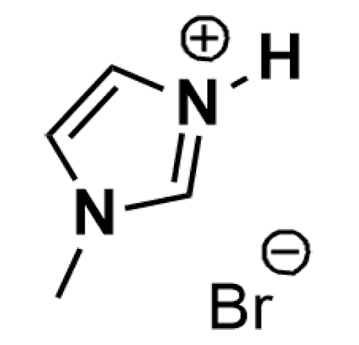
1-Methylimidazolium bromide, >98%
Price range: $99.01 through $763.73
Product Code: IL-0267-SGCAS NO: 101023-58-9
- Chemical Formula: C4H7BrN2
- Synonyms: MIM Br, 1-Methylimidazole Hydrobromide
- Aromatic
- Hydrophilic
- Protic
To order 5kg or more, please email us at info@roco.global
**This product will incur a $97.00 HazMat fee when the order is placed.**
SUM Formula: C4H7BrN2
Molecular Weight: 163.02
Melting Point: 48 °C
Density: 1.292 g/cm
Purity: >98%
- SUM Formula: C4H7BrN2
- Molecular Weight: 163.02
- Melting Point: 48 °C
- Density: 1.292 g/cm
1-Methylimidazolium bromide, CAS: 101023-58-9
Key Application:
1. Precursor for Tailored Ionic Liquid Synthesis
- Serves as a key quaternization intermediate for producing a wide range of 1‑alkyl‑3‑methylimidazolium ionic liquids via N‑alkylation.
- Enables modular synthesis of ILs with tunable hydrophobicity, viscosity, conductivity, and thermal stability.
- Commonly used in research settings to generate chloride, tetrafluoroborate, PF₆⁻, NTf₂⁻, FSI⁻, and other anion‑exchanged ILs.
2. Building Block for Functional Organic Salts
- Acts as a versatile imidazolium cation source for preparing functional salts used in:
- Organocatalysis
- Phase‑transfer catalysis
- Electrolyte additives
- Polymeric ionic liquids (PILs)
- Supports synthesis of IL monomers for ion‑conducting polymer networks and membrane materials.
3. Catalyst and Co‑Catalyst Component
- Used as a cationic scaffold in catalytic systems where imidazolium species stabilize reactive intermediates.
- Plays a role in:
- Transition‑metal catalysis (e.g., Pd, Ru, Ni systems)
- CO₂ activation studies
- N‑heterocyclic carbene (NHC) precursor chemistry
4. Solvent and Microenvironment Modifier
- Introduced in small quantities to tune polarity, ionic strength, or microviscosity in:
- Organic synthesis
- Supramolecular assembly
- Biomolecular stabilization studies
- Useful in screening experiments where the imidazolium cation’s hydrogen‑bonding profile influences reaction outcomes.
5. Electrochemical and Conductive Materials Research
- Serves as a precursor for ILs used in:
- Electrolytes for batteries and supercapacitors
- Electrodeposition baths
- Ion‑transport membranes
- The bromide form is particularly useful for anion‑exchange reactions that yield high‑conductivity ionic liquids.
6. Surfactant and Self‑Assembly Studies
- Imidazolium salts are widely used to probe:
- Micelle formation
- Interfacial charge effects
- Nanostructured soft materials
- 1‑Methylimidazolium bromide provides a short‑chain, low‑hydrophobicity benchmark for comparative studies.
7. Template and Structure‑Directing Agent
- Employed in the synthesis of:
- Zeolites and mesoporous materials
- Metal–organic frameworks (MOFs)
- Nanoparticle assemblies
- The compact cation size allows fine control over pore geometry and crystallization pathways.
Please contact us if you want to learn more or need assistance with your order.