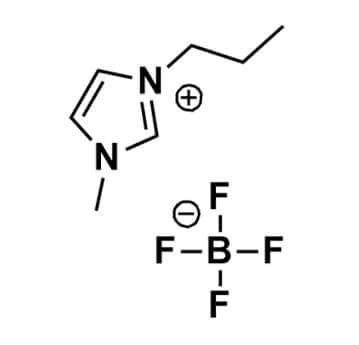
1-Methyl-3-propylimidazolium tetrafluoroborate, >98%
Price range: $121.62 through $6,971.12
Product Code: IL-0143-HPCAS NO: 244193-48-4
- Chemical Formula: C7H13BF4N2
- Synonyms: PMIM BF4, C1C3Im BF4, Im13 BF4
- Anion decomposes slowly in the presence of water
- Hydrophilic
- Aromatic
Conductivity: 4.080 mS/cm
SUM Formula: C7H13BF4N2
Molecular Weight: 212.0
Melting Point: -47 °C
Density: 1.27 g/cm³ (20 °C)
Purity: >98%
Viscosity: 74.4 cP (25 °C)
- SUM Formula: C7H13BF4N2
- Molecular Weight: 212.0
- Melting Point: -47 °C
- Density: 1.27 g/cm³ (20 °C)
- Viscosity: 74.4 cP (25 °C)
1-Methyl-3-propylimidazolium tetrafluoroborate, CAS: 244193-48-4
Key Applications:
Solvent and Electrolyte Functions
- Serves as a low‑volatility, thermally stable solvent for organic synthesis, particularly in substitution, cyclization, and transition‑metal‑catalyzed reactions.
- Functions as an electrolyte component in electrochemical systems requiring moderate viscosity and stable ionic conductivity.
- Supports electrodeposition studies where BF₄⁻ anions promote smooth metal film formation and minimize passivation.
Catalysis and Reaction Media
- Provides a tunable, non‑aqueous medium for acid‑catalyzed transformations, including esterifications and alkylations.
- Enhances selectivity in biphasic and phase‑transfer systems due to its balanced polarity and miscibility profile.
- Useful in microwave‑assisted synthesis, where ionic liquids accelerate heating and improve reaction efficiency.
Separation and Extraction
- Employed in liquid–liquid extraction of aromatic hydrocarbons, phenolics, and metal ions, leveraging BF₄⁻’s relatively weak coordinating behavior.
- Supports chromatographic method development as an additive or stationary‑phase modifier to improve peak shape and analyte retention control.
Materials and Polymer Science
- Acts as a plasticizing and conductivity‑enhancing additive in polymer electrolytes, ion gels, and composite membranes.
- Used in templating and dispersion of nanoparticles, where the imidazolium cation stabilizes metal or metal‑oxide surfaces.
- Facilitates the preparation of hybrid materials requiring ionic domains for charge transport or structural tuning.
Spectroscopy and Analytical Chemistry
- Provides a stable, low‑volatility matrix for spectroscopic studies of solvation, ion pairing, and reaction intermediates.
- Serves as a calibration or reference medium in electroanalytical experiments due to its predictable ionic mobility.
Energy and Environmental Applications
- Investigated as a component in redox‑flow and dye‑sensitized solar cell electrolytes where BF₄⁻ offers oxidative stability.
- Supports CO₂ capture and gas‑solubility studies, particularly in systems evaluating cation structure–property relationships.
Please contact us if you want to learn more or if you need assistance with your order.