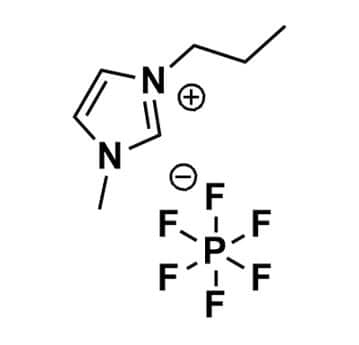
1-Methyl-3-propylimidazolium hexafluorophosphate, >99%
Price range: $142.89 through $8,209.56
Product Code: IL-0142-HPCAS NO: 216300-12-8
- Chemical Formula: C7H13F6N2P
- Synonyms: PMIM PF6, C1C3Im PF6, Im13 PF6
- Anion decomposes slowly in the presence of water.
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
SUM Formula: C7H13F6N2P
Molecular Weight: 270.16
Melting Point: 41 °C
Density: 1.518 g/cm³
Purity: >99%
- SUM Formula: C7H13F6N2P
- Molecular Weight: 270.16
- Melting Point: 41 °C
- Density: 1.518 g/cm³
1-Methyl-3-propylimidazolium hexafluorophosphate, CAS: 216300-12-8
Key Applications:
Electrochemical Systems
- Serves as a high‑conductivity ionic medium in electrochemical cells where low viscosity and stable PF₆⁻ coordination are required.
- Used as a supporting electrolyte in cyclic voltammetry, chronoamperometry, and electrodeposition studies due to its wide electrochemical window.
- Enhances ion transport in prototype supercapacitors and redox‑flow testbeds evaluating imidazolium‑based electrolytes.
Catalysis and Synthesis
- Functions as a reaction medium for transition‑metal‑catalyzed couplings, alkylations, and oxidations where non‑coordinating anions are preferred.
- Supports biphasic and phase‑transfer catalysis, enabling improved separation and catalyst recyclability.
- Applied in selective organic transformations requiring thermally stable, low‑volatility solvents.
Separation Science
- Utilized in liquid–liquid extraction systems for metal ions, aromatic hydrocarbons, and polar organics, leveraging its tunable polarity and hydrophobic PF₆⁻ anion.
- Employed in chromatographic method development as an ionic additive to modify retention behavior and improve peak resolution.
Materials and Polymer Processing
- Acts as a structuring agent or plasticizer in polymer electrolytes, ionogels, and composite membranes.
- Supports nanoparticle dispersion and stabilization in colloidal systems, particularly for metal and metal‑oxide nanomaterials.
- Used in templating and self‑assembly studies where ionic liquid microenvironments influence morphology.
Thermal and Green‑Chemistry Applications
- Suitable for high‑temperature reaction environments due to its low vapor pressure and thermal stability.
- Enables solvent‑minimized or solvent‑free processing in sustainable synthesis workflows.
Analytical and Spectroscopic Uses
- Provides a low‑volatility matrix for spectroscopic measurements, including NMR and IR studies of solute–ion interactions.
- Serves as a calibration or reference medium in studies comparing PF₆⁻‑based ionic liquids across alkyl‑chain homologues.
Please contact us if you want to learn more or need assistance with your order.