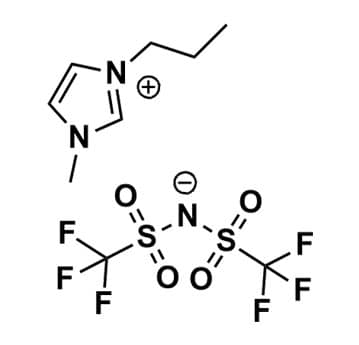
1-Methyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide, >99.5%
Price range: $193.81 through $2,655.39
Product Code: IL-0024-UPCAS NO: 216299-72-8
- Chemical Formula: C9H13F6N3O4S2
- Synonyms: PMIM BTA, PMIM NTf2, PMIM TFSI, PMIM BTI, C1C3Im BTA, Im13 BTA
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
For High-Purity of >99% refer to IL-0024-HP
Conductivity: 4.40 mS/cm (20 °C)
SUM Formula: C9H13F6N3O4S2
Molecular Weight: 405.34
Melting Point: -38 °C
Density: 1.48 g/cm³ (18 °C)
ECW: 4.9 V
Purity: >99.5%
Viscosity: 56 cP (19 °C)
- SUM Formula: C9H13F6N3O4S2
- Molecular Weight: 405.34
- Melting Point: -38 °C
- Density: 1.48 g/cm³ (18 °C)
- ECW: 4.9 V
- Viscosity: 56 cP (19 °C)
1-Methyl-3-propylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 216299-72-8
Key Applications:
Electrochemical Systems and High‑Performance Lithium Metal Batteries
- Serves as a non‑volatile, non‑flammable electrolyte component for lithium metal batteries, improving safety and cycle life.
- Enhances ionic conductivity and SEI stability in high‑energy‑density systems used in electric vehicles and renewable‑energy storage infrastructure.
- Compatible with high‑voltage cathode chemistries due to the wide electrochemical stability window of the TFSI anion.
- Reduces dendrite formation on lithium metal surfaces, supporting next‑generation solid‑state and hybrid electrolyte designs.
Phenolic Compound Extraction and Water Purification
- Selectively extracts phenolic pollutants (phenol, cresols, chlorophenols) from industrial wastewater streams.
- Hydrophobicity and tunable solvation enable high distribution coefficients and efficient phase separation.
- Supports multi‑cycle extraction processes with minimal degradation, making it suitable for continuous purification systems.
- Useful in advanced water‑treatment modules where low volatility and chemical stability are required.
Azeotropic and Difficult Mixture Separation
- Breaks azeotropes by altering relative volatilities through strong, selective solvation of target components.
- Effective in separating alcohol–water, aromatic–aliphatic, and other industrially relevant azeotropic systems.
- Enables energy‑efficient extractive distillation, reducing thermal load and improving separation purity.
- Useful in solvent recovery, fine‑chemical purification, and biomass‑derived feedstock upgrading.
Catalysis and Reaction Media
- Provides a stable, non‑volatile medium for acid‑, base‑, and metal‑catalyzed transformations.
- Enhances selectivity in alkylation, oxidation, and coupling reactions due to strong ion–dipole interactions.
- Supports biphasic catalysis where easy product–catalyst separation is required.
Materials Science and Polymer Processing
- Acts as a plasticizer or conductivity enhancer in polymer electrolytes, ion gels, and flexible electronics.
- Useful in dispersing nanomaterials such as graphene, CNTs, and metal oxides due to strong π–cation interactions.
- Improves thermal stability and ionic mobility in advanced composite materials.
Gas Absorption and Separation
- Demonstrates selective solubility for gases such as SO₂, CO₂, and H₂S.
- Supports membrane‑based separations and gas‑capture systems where chemical stability is essential.
Tribology and High‑Temperature Lubrication
- Functions as a high‑temperature, low‑volatility lubricant or lubricant additive.
- Reduces wear and friction in metal–metal and metal–ceramic interfaces under extreme conditions.
Electrodeposition and Surface Engineering
- Provides controlled metal deposition environments for Cu, Ni, and noble metals.
- Enables smooth, uniform coatings with reduced dendritic growth.
Please contact us if you want to learn more or need assistance with your order.