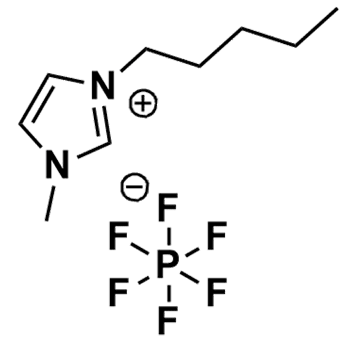
1-Methyl-3-pentylimidazolium hexafluorophosphate, >99%
Price range: $178.24 through $10,311.55
Product Code: IL-0306-HPCAS NO: 280779-52-4
- Chemical Formula: C9H17F6N2P
- Synonyms: PentMIM PF6 , C 1 C 5 Im PF6, Im 15 PF 6
- Anion decomposes slowly in the presence of water
- Weakly-coordination Anion
- Hydrophobic
- Armoatic
SUM Formula: C9H17F6N2P
Molecular Weight: 298.21
Melting Point: <RT
Purity: >99%
- SUM Formula: C9H17F6N2P
- Molecular Weight: 298.21
- Melting Point:
1-Methyl-3-pentylimidazolium hexafluorophosphate, CAS: 280779-52-4
Key Applications
Catalysis and Reaction Media
- Serves as a non‑volatile solvent for transition‑metal catalysis, enabling improved selectivity and recyclability of catalytic systems.
- Supports biphasic and multiphasic reaction designs where hydrophobic ionic liquids enhance product separation.
- Provides a stable medium for organometallic transformations sensitive to moisture or conventional organic solvents.
Electrochemical Systems
- Functions as an electrolyte component in electrochemical cells requiring wide electrochemical windows and low vapor pressure.
- Useful in studies of ion transport, electrode interfaces, and ionic‑liquid‑based capacitors.
- Supports development of ionic‑liquid‑modified electrodes for sensing and analytical applications.
Extraction and Separation
- Enables liquid–liquid extraction of metal ions, organic compounds, and hydrophobic analytes due to its immiscibility with water and tunable solvation behavior.
- Applied in separation workflows for aromatic compounds, dyes, and environmental contaminants.
- Suitable for designing task‑specific extraction systems where PF₆⁻ anions contribute to phase stability.
Materials Science and Polymer Processing
- Acts as a structuring agent or plasticizer in polymer synthesis and processing, influencing conductivity, morphology, and mechanical properties.
- Supports fabrication of ion‑conductive films, membranes, and composite materials.
- Useful in templating studies for nanoporous materials and ionic‑liquid‑assisted self‑assembly.
Spectroscopy and Fundamental Studies
- Provides a controlled, low‑volatility environment for NMR, IR, and UV‑Vis investigations of solvation, ion pairing, and molecular dynamics.
- Used in benchmarking studies comparing alkyl‑chain effects across imidazolium PF₆ ionic liquids.
- Supports mechanistic research on ion–solvent interactions and phase behavior.
Please contact us if you want to learn more or need assistance with your order.