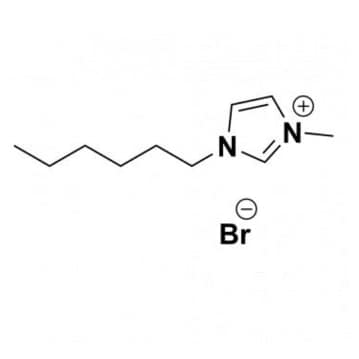
1-Methyl-3-pentylimidazolium bromide, >99%
Price range: $190.97 through $8,124.78
Product Code: IL-0297-HPCAS NO: 343851-31-0
- Chemical Formula: C9H17BrN2
- Synonyms: PentMIM Br, C1 C5 Im Br, Im 15 Br
- Aromatic
SUM Formula: C9H17BrN2
Molecular Weight: 233.15
Melting Point: <RT
Purity: >99%
- SUM Formula: C9H17BrN2
- Molecular Weight: 233.15
- Melting Point:
1-Methyl-3-pentylimidazolium bromide, CAS: 343851-31-0
Key Applications:
Solvent and Reaction Medium
- Functions as a polar, non‑volatile ionic liquid suitable for organic synthesis requiring controlled solvation and reduced vapor pressure.
- Supports acid‑ and base‑catalyzed transformations, including alkylation, esterification, and nucleophilic substitution under mild conditions.
- Provides tunable microenvironments for homogeneous catalysis, improving selectivity in transition‑metal‑mediated reactions.
Phase‑Transfer and Extraction Processes
- Acts as an efficient phase‑transfer medium for biphasic organic–aqueous systems, enhancing mass transfer and reaction rates.
- Useful in selective extraction of metal ions, dyes, and organic contaminants due to its amphiphilic cation and halide counterion.
Electrochemical and Materials Applications
- Serves as a stable ionic conductor in electrochemical studies, including electrodeposition and redox‑active material testing.
- Can be incorporated into polymer electrolytes or ion‑conductive films to improve ionic mobility and thermal stability.
- Supports templating and dispersion of nanomaterials, including metal nanoparticles and carbon‑based nanostructures.
Catalysis and Green Chemistry
- Provides a recyclable medium for catalytic cycles, reducing solvent waste and improving process sustainability.
- Enhances catalytic activity of metal complexes and organocatalysts by stabilizing reactive intermediates.
Biomolecular and Analytical Uses
- Useful as a solubilizing agent for hydrophobic or partially hydrophobic biomolecules in analytical workflows.
- Can modulate enzyme activity or protein stability in controlled studies due to its tunable polarity and ionic strength.
Surfactant‑Like and Self‑Assembly Behavior
- The C5 alkyl chain imparts moderate hydrophobicity, enabling micelle‑like aggregation in aqueous or mixed‑solvent systems.
- Supports formation of structured phases useful in templating, encapsulation, and controlled‑release research.
Please contact us if you want to learn more or need assistance with your order.