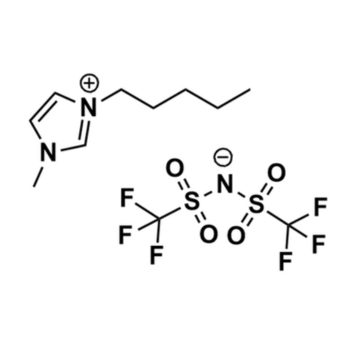
1-Methyl-3-pentylimidazolium bis(trifluoromethylsulfonyl)imide, >99%
Price range: $268.84 through $14,804.30
Product Code: IL-0300-HPCAS NO: 280779-53-5
- Chemical Formula: C11H17F6N3O4S2
- Synonyms: PentMIM BTA, PentMIM NTf 2, PentMIM TFSI, PentMIM BTI, C 1 C 5 Im BTA, Im 15 BTA
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 3.28 mS/cm (30°C)
SUM Formula: C11H17F6N3O4S2
Molecular Weight: 433.39
Melting Point: <RT
Purity: >99%
Viscosity: 59.3 cP (25 °C)
- SUM Formula: C11H17F6N3O4S2
- Molecular Weight: 433.39
- Melting Point:
- Viscosity: 59.3 cP (25 °C)
1-Methyl-3-pentylimidazolium bis(trifluoromethylsulfonyl)imide, CAS: 280779-53-5
Key Applications:
Electrochemical & Energy Applications
- Electrolytes for high‑voltage lithium and sodium systems, leveraging the NTf₂⁻ anion’s wide electrochemical window and thermal stability.
- Ion‑conductive media in supercapacitors, where moderate viscosity supports improved ion mobility compared to longer‑chain homologues.
- Redox‑active electrolyte formulations for flow batteries and metal–air systems.
Catalysis & Synthesis
- Reaction media for transition‑metal catalysis, including C–C coupling, hydrogenation, and oxidation reactions requiring non‑volatile, thermally robust solvents.
- Phase‑transfer catalysis, where the C₅ chain provides tunable hydrophobicity for biphasic organic–aqueous systems.
- Stabilization of reactive intermediates in organometallic and heterocycle synthesis.
Materials Science & Polymer Processing
- Solvent and plasticizer for engineering polymers, including polyimides, polyacrylates, and fluorinated materials.
- Template or structure‑directing agent in nanoparticle synthesis and porous material fabrication.
- Processing aid for conductive composites, improving dispersion of carbon nanotubes, graphene, and metal oxides.
Separation & Extraction
- Selective extraction of aromatic hydrocarbons, sulfur species, and metal ions due to strong π‑interaction and coordination capability.
- CO₂ capture and gas separation media, benefiting from NTf₂⁻’s low coordinating nature and the cation’s moderate hydrophobicity.
- Liquid–liquid extraction in bioprocessing, including purification of bioactive compounds and specialty organics.
Lubrication & Surface Engineering
- High‑temperature, low‑volatility lubricant additive for metal–metal and metal–ceramic interfaces.
- Anti‑wear and friction‑reducing films, where the ionic liquid forms stable boundary layers under load.
- Surface treatment agent for corrosion‑resistant coatings.
Analytical & Laboratory Uses
- Non‑aqueous mobile phase modifier in chromatography for enhanced separation of hydrophobic analytes.
- Matrix or dispersive medium in spectroscopic and electroanalytical methods requiring low vapor pressure and high thermal stability.
- Solvent for crystallization studies of organics and coordination complexes.
Please contact us if you want to learn more or need assistance with your order.