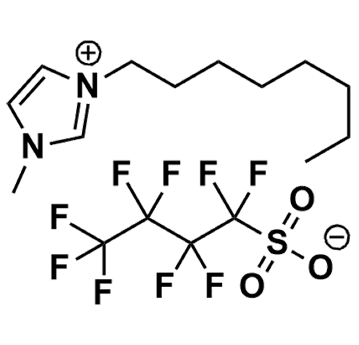
1-Methyl-3-octylimidazolium perfluorobutanesulfonate, >99%
Price range: $226.45 through $13,155.34
Product Code: IL-0312-HPCAS NO: 905972-83-0
- Chemical Formula: C16H23F9N2O3S
- Synonyms: OMIM PFBS, C1C8Im PFBS, OMIM ONf, 1-Methyl-3-octylimidazolium nonafluorobutanesulfonate, 1-Methyl-3-octylimidazolium nonaflate
- Weakly-coordinating anion
- Hydrophobic
- Aromatic
Conductivity: 0.46 mS/cm (40 °C)
SUM Formula: C16H23F9N2O3S
Molecular Weight: 494.42
Melting Point: 35 °C
Purity: >99%
Viscosity: 171 cP (40 °C)
- SUM Formula: C16H23F9N2O3S
- Molecular Weight: 494.42
- Melting Point: 35 °C
- Viscosity: 171 cP (40 °C)
1-Methyl-3-octylimidazolium perfluorobutanesulfonate, CAS: 905972-83-0
Key Applications:
Electrochemical Systems
- Serves as a hydrophobic, wide‑electrochemical‑window electrolyte component for high‑voltage supercapacitors and advanced battery chemistries.
- Enhances interfacial stability in electrode–electrolyte systems due to the bulky, weakly coordinating perfluorobutanesulfonate anion.
- Supports stable ion transport in mixed‑solvent or solvent‑free ionic liquid electrolytes for long‑cycle‑life devices.
Separation Science and Extraction
- Functions as a tunable, non‑volatile extraction medium for hydrophobic organics, fluorinated compounds, and metal complexes.
- Useful in biphasic ionic liquid–aqueous systems where low miscibility and high thermal stability are required.
- Enables selective partitioning in liquid–liquid extraction workflows for analytical and industrial separations.
Catalysis and Reaction Media
- Provides a thermally robust, non‑flammable reaction medium for acid‑catalyzed, transition‑metal‑catalyzed, and fluorophilic transformations.
- Improves catalyst lifetime and turnover in systems sensitive to moisture or coordinating anions.
- Supports phase‑separated catalysis where product isolation is simplified by the ionic liquid’s hydrophobicity.
Materials Processing and Surface Engineering
- Acts as a structuring agent or solvent for fluorinated polymers, coatings, and surface‑modification processes.
- Facilitates deposition and patterning of hydrophobic or low‑surface‑energy materials.
- Useful in nanoparticle synthesis where controlled nucleation and growth benefit from the ionic liquid’s low volatility and high ionic strength.
Spectroscopy and Analytical Chemistry
- Provides a stable, low‑vapor‑pressure matrix for spectroscopic studies of fluorinated species and ion–solvent interactions.
- Suitable as a reference medium for probing solvation dynamics in hydrophobic ionic environments.
Thermal and Chemical Stability Applications
- Employed in high‑temperature processes requiring non‑flammable, chemically inert ionic media.
- Supports heat‑transfer or thermal‑management research where low volatility and high decomposition thresholds are essential.
Please contact us if you want to learn more or need assistance with your order.