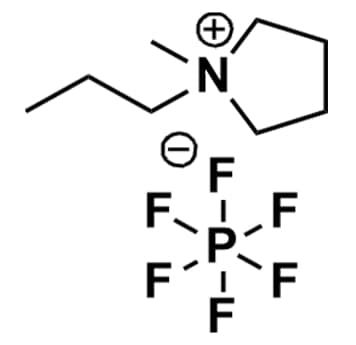
1-Methyl-1-propylpyrrolidinium hexafluorophosphate, >99%
Price range: $284.42 through $12,528.26
Product Code: IL-0148-HPCAS NO: 327022-58-2
- Chemical Formula: C8H18F6NP
- Synonyms: PMPyrr PF6, PYR13 PF6, PY13 PF6, N-Methyl-N-propylpyrrolidinium hexafluorophosphate
- Anion decomposes slowly in the presence of water
- Weakly-coordinating anion
- Hydrophobic
SUM Formula: C8H18F6NP
Molecular Weight: 273.20
Melting Point: 112 °C
Purity: >99%
- SUM Formula: C8H18F6NP
- Molecular Weight: 273.20
- Melting Point: 112 °C
1-Methyl-1-propylpyrrolidinium hexafluorophosphate, CAS: 327022-58-2
Key Applications:
Electrochemical Systems & Energy Storage
- Employed as a non‑aqueous electrolyte component in supercapacitors and high‑voltage electrochemical cells requiring wide electrochemical windows.
- Used to formulate ionic‑liquid‑based electrolytes with enhanced thermal stability and reduced volatility for next‑generation battery research.
- Serves as a conductivity‑enhancing additive in redox‑active organic systems and ionic‑liquid‑gel polymer electrolytes.
Solvent & Extraction Applications
- Functions as a hydrophobic extraction medium for metal ions, organometallic complexes, and hydrophobic organic solutes.
- Applied in liquid–liquid extraction systems where PF₆⁻ ionic liquids provide tunable partitioning behavior and low miscibility with water.
- Utilized in supported ionic liquid phases (SILPs) for separations and catalytic flow processes.
Catalysis & Reaction Media
- Acts as a non‑volatile, thermally stable solvent for transition‑metal catalysis, including hydrogenation, coupling, and oxidation reactions.
- Supports biphasic catalysis, enabling catalyst recovery and reuse due to phase immiscibility with many organic solvents.
- Provides a controlled ionic environment for organocatalytic and acid‑base‑mediated transformations.
Materials Science & Polymer Engineering
- Used as a structuring agent in polymer electrolytes, ion gels, and composite materials requiring ionic conductivity and mechanical stability.
- Incorporated into nanomaterial synthesis, including templated nanoparticle formation and surface functionalization.
- Serves as a plasticizer or ionic mobility enhancer in specialty polymer matrices.
Spectroscopy, Transport, and Fundamental Studies
- Selected for ionic transport, viscosity, and ion‑pairing studies due to its well‑defined cation structure and hydrophobic PF₆⁻ anion.
- Used in NMR, Raman, and IR investigations of ion dynamics, solvation, and electrochemical interfaces.
- Supports computational benchmarking and experimental validation of ion–solvent interaction models.
Please contact us if you want to learn more or need assistance with your order.