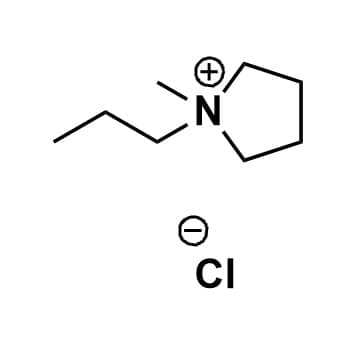
1-Methyl-1-propylpyrrolidinium chloride, >99%
Price range: $152.77 through $4,911.65
Product Code: IL-0146-HPCAS NO: 528818-82-8
- Chemical Fr.: C8H18ClN
- Synonyms: PMPyrr Cl, PYR13 Cl, PY13 Cl, N-Methyl-N-propylpyrrolidinium chloride
- Hydrophilic
SUM Formula: C8H18ClN
Molecular Weight: 163.69
Melting Point: 236 °C
Density: 1.132 g/cm³
Purity: >99%
- SUM Formula: C8H18ClN
- Molecular Weight: 163.69
- Melting Point: 236 °C
- Density: 1.132 g/cm³
1-Methyl-1-propylpyrrolidinium chloride, CAS: 528818-82-8
Key Applications:
Electrochemistry & Energy Storage
- Employed as an ionic liquid precursor or supporting electrolyte in aqueous and mixed‑solvent electrochemical systems.
- Enhances ionic conductivity and electrode interfacial stability in redox‑flow, metal‑ion, and hybrid aqueous battery research.
- Used in electrodeposition studies where chloride coordination influences metal nucleation and morphology.
Catalysis & Reaction Media
- Serves as a polar, non‑volatile medium for transition‑metal catalysis, phase‑transfer catalysis, and halide‑assisted transformations.
- Supports selective organic transformations where chloride activity modulates reaction pathways.
- Utilized in mechanistic studies probing ion–pairing, solvation, and microenvironment effects.
Polymer & Materials Science
- Acts as a structure‑directing additive in polymer electrolytes, ion‑conducting membranes, and block‑copolymer assemblies.
- Incorporated into ionogels and hybrid materials to tune mechanical strength, ionic mobility, and thermal stability.
- Used in templating and self‑assembly studies where pyrrolidinium cations influence nanoscale ordering.
Solvent Extraction & Separation Science
- Applied in liquid–liquid extraction systems for selective metal ion coordination and chloride‑driven speciation control.
- Supports separation processes requiring high polarity, low volatility, and tunable ion‑pair interactions.
Spectroscopy & Fundamental Studies
- Used as a model pyrrolidinium ionic liquid for NMR, IR, Raman, and dielectric spectroscopy to probe ion dynamics, hydrogen bonding, and solvation structure.
- Supports computational and experimental benchmarking of cation–anion interactions in chloride‑based ionic liquids.
Please contact us if you want to learn more or need assistance with your order.