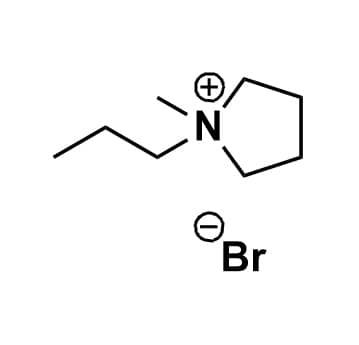
1-Methyl-1-propylpyrrolidinium bromide, >99%
Product Code: RIL-0145-HPCAS NO: 608140-09-6
- Chemical Formula: C8H18BrN
- Synonyms: PMPyrr Br, PYR13 Br, PY13 Br, N-Methyl-N-propylpyrrolidinium bromide
- Hydrophilic
This is a custom synthesis product. Price upon request
Please email us at info@roco.global to place an order
SUM Formula: C8H18BrN
Molecular Weight: 208.14
Melting Point: 210 °C
Density: 1.342 g/cm³
Purity: >99%
- SUM Formula: C8H18BrN
- Molecular Weight: 208.14
- Melting Point: 210 °C
- Density: 1.342 g/cm³
1-Methyl-1-propylpyrrolidinium bromide, CAS: 608140-09-6
Key Applications
Electrochemical and Energy Applications
- Employed as a supporting electrolyte or ionic additive in electrochemical studies requiring stable, non‑reactive cations.
- Useful in evaluating ion‑transport behavior in prototype ionic liquid systems and mixed‑solvent electrolytes.
- Supports charge‑transfer studies, electrode interface characterization, and conductivity benchmarking.
Materials Science and Polymer Systems
- Incorporated into polymer matrices to tune ionic conductivity, plasticization behavior, and mechanical properties.
- Used in the preparation of ion‑conducting films, membranes, and composite materials for research applications.
- Serves as a structure‑directing ion in templated materials and organized soft‑matter assemblies.
Catalysis and Synthetic Chemistry
- Functions as a phase‑transfer catalyst or ionic medium in reactions requiring enhanced solubility of inorganic species.
- Provides a controlled ionic environment for nucleophilic substitutions, alkylations, and other transformations sensitive to solvent polarity.
- Supports mechanistic studies where quaternary ammonium ions are used as benchmarks or reference cations.
Analytical and Separation Science
- Utilized as an ion‑pairing reagent in chromatography to improve retention and separation of analytes with complementary charge profiles.
- Applied in method development for ion‑exchange and electrophoretic systems requiring stable, well‑defined cations.
General Research and Formulation Uses
- Serves as a precursor or reference compound in the development of new ionic liquids and quaternary ammonium derivatives.
- Useful in solvation, miscibility, and thermodynamic studies involving bromide salts and pyrrolidinium‑based ions.
- Supports formulation screening where cation identity influences viscosity, polarity, or interfacial behavior.
Please contact us if you want to learn more or need assistance with your order.