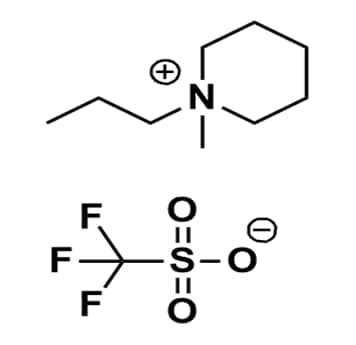
1-Methyl-1-propylpiperidinium triflate, >98%
Price range: $366.49 through $4,116.09
Product Code: IL-0175-HPCAS NO: 1221445-34-6
- Chemical Formula: C10H20F3NO3S
- Synonyms: N-Methyl-N-propylpiperidinium triflate, 1-Methyl-1-propylpiperidinium trifluoromethanesulfonate, PMPip OTf, PIP13 OTf
SUM Formula: C10H20F3NO3S
Molecular Weight: 291.33
Melting Point: 71 °C
Purity: >98%
- SUM Formula: C10H20F3NO3S
- Molecular Weight: 291.33
- Melting Point: 71 °C
1-Methyl-1-propylpiperidinium triflate, CAS: 1221445-34-6
Key Applications:
Electrochemical Systems
- Serves as a stable ionic liquid cation paired with a weakly coordinating triflate anion, enabling high ionic conductivity in non‑aqueous media.
- Used as a supporting electrolyte in voltammetry, electrodeposition studies, and redox‑active material screening.
- Suitable for high‑temperature electrochemical environments due to its thermal and electrochemical stability.
Battery and Energy Storage Research
- Functions as a component in prototype electrolyte formulations for lithium, sodium, and multivalent systems where low volatility and wide electrochemical windows are required.
- Supports studies on ion transport, SEI formation, and solvent–salt interactions in next‑generation battery chemistries.
Catalysis and Organic Synthesis
- Acts as a non‑volatile, thermally robust reaction medium for acid‑sensitive or moisture‑sensitive transformations.
- Useful in biphasic catalysis, transition‑metal‑mediated reactions, and systems requiring tunable polarity without introducing coordinating impurities.
Materials Science and Polymer Processing
- Employed as a plasticizing or ion‑conducting additive in polymer electrolytes, ionogels, and hybrid organic–inorganic materials.
- Enhances ionic mobility and mechanical flexibility in solid‑state matrices.
Spectroscopy and Fundamental Studies
- Provides a well‑defined ionic environment for NMR, IR, and conductivity studies examining ion pairing, solvation, and transport mechanisms.
- Used in benchmarking structure–property relationships across piperidinium‑based ionic liquids.
Separation Science
- Supports liquid–liquid extraction and phase‑transfer processes where hydrophobicity, low vapor pressure, and controlled acidity are advantageous.
- Useful in selective metal ion extraction and purification workflows.
Please contact us if you would like to learn more or if you require assistance with your order.